Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Glentham Life Sciences | UK | |||
|---|---|---|---|---|
 | www.glenthamls.com | |||
 | +44 (2033) 978-798 | |||
 | +44 (2033) 978-909 | |||
 | info@glenthamls.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Typu Group | China | |||
|---|---|---|---|---|
 | www.typugroup.com | |||
 | +86 85220966 | |||
 | order@typugroup.com | |||
| Chemical distributor since 2008 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Generay Biotech (Shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.generay.com.cn | |||
 | +86 (21) 6762-6003 6762-6050 6762-6120 | |||
 | +86 (21) 6762-6011 | |||
 | order@generay.com.cn | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| SBS Genetech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sbsbio.com | |||
 | +86 (10) 8278-4296 8278-4292 6296-9345 | |||
 | +86 (10) 8278-4290 | |||
 | info@sbsbio.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Classification | API >> Antibiotics >> Polyene |
|---|---|
| Name | Amphotericin B |
| Synonyms | 33-[(3-Amino-3,6-dideoxy-beta-D-mannopyranosyl)oxy]-1,3,5,6,9,11,17,37-octahydroxy-15,16,18-trimethyl-13-oxo-14,39-Dioxabicyclo[33.3.1]nonatriaconta-19,21,23,25,27,29,31-heptaene-36-carboxylic acid; Fungizone |
| Molecular Structure | 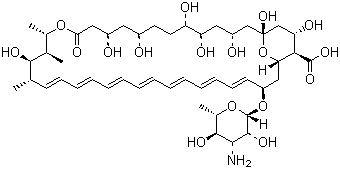 |
| Molecular Formula | C47H73NO17 |
| Molecular Weight | 924.08 |
| CAS Registry Number | 1397-89-3 |
| EC Number | 215-742-2 |
| SMILES | C[C@H]1/C=C/C=C/C=C/C=C/C=C/C=C/C=C/[C@@H](C[C@H]2[C@@H]([C@H](C[C@](O2)(C[C@H](C[C@H]([C@@H](CC[C@H](C[C@H](CC(=O)O[C@H]([C@@H]([C@@H]1O)C)C)O)O)O)O)O)O)O)C(=O)O)O[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)O)N)O |
| Solubility | <0.1 g/100 mL (21 °C, water) |
|---|---|
| Density | 1.34±0.1 g/cm3 |
| Index of Refraction | 1.614, Calc.* |
| Boiling Point | 1140.4±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 643.5±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P271-P280-P302+P352-P304+P340-P305+P351+P338-P312-P362-P403+P233-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Discovered in 1955 by researchers Rachel Fuller Brown and Elizabeth Lee Hazen at the New York State Department of Health, Amphotericin B emerged from their pioneering work in antifungal research. The breakthrough came through systematic screening of soil samples for potential antifungal activity. The compound was isolated from cultures of Streptomyces nodosus, a bacterium found in soil. Amphotericin B remains a cornerstone in the treatment of severe systemic fungal infections. It is highly effective against a broad spectrum of fungal pathogens, including Candida and Aspergillus species. Its mechanism of action involves binding to ergosterol, a key component of fungal cell membranes, leading to disruption of membrane integrity and subsequent cell death. This makes it particularly useful in treating life-threatening fungal infections, such as invasive candidiasis and aspergillosis, especially in immunocompromised patients. Amphotericin B is also used in the treatment of fungal meningitis, a serious infection of the central nervous system caused by fungi such as Cryptococcus neoformans. Intrathecal or intraventricular administration of the drug can achieve therapeutic concentrations in the cerebrospinal fluid, providing effective treatment against fungal pathogens within the central nervous system. In certain high-risk patient populations, such as those undergoing bone marrow transplantation or receiving high-dose chemotherapy, prophylactic use of Amphotericin B may be employed to prevent invasive fungal infections. This proactive approach helps reduce the incidence of fungal infections, which can significantly impact patient outcomes in immunocompromised individuals. Amphotericin B is also used in the treatment of leishmaniasis, a parasitic disease caused by protozoa of the Leishmania genus. References 1979. Interaction of the polyene antibiotic amphotericin B with phospholipid bilayer membranes: A circular dichroism study. Biochemical and Biophysical Research Communications. DOI: 10.1016/0006-291x(79)91507-9 1979. Treatment of Experimental Naegleria Meningoencephalitis with a Combination of Amphotericin B and Rifamycin. Scandinavian Journal of Infectious Diseases. DOI: 10.3109/inf.1979.11.issue-2.10 1979. Comparison of Microdilution and Broth Dilution Techniques for the Susceptibility Testing of Yeasts to 5-Fluorocytosine and Amphotericin B. Antimicrobial Agents and Chemotherapy. DOI: 10.1128/aac.15.3.475 |
| Market Analysis Reports |