Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Boron Molecular Inc. | USA | |||
|---|---|---|---|---|
 | www.boronmolecular.com | |||
 | +1 (919) 981-4441 | |||
 | +1 (919) 981-4442 | |||
 | sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Wuxi Southward Chemical Auxiliary Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nanfangchem.com | |||
 | +86 (25) 8479-5611 | |||
 | +86 (510) 8558-1040 | |||
 | nanfangchem@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Bondchemistry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bondchemistry.com | |||
 | +86 15858185835 | |||
 | sales@bondchemistry.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Hike Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njhkchem.com | |||
 | +86 (25) 5211-0956 | |||
 | +86 (25) 5211-0956 | |||
 | sophia@njhkchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17372769668 | |||
 | WhatsApp:17372769668 | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Syn-Finechem Laboratories Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.synfinechem.com | |||
 | +91 (40) 2309-5094 | |||
 | +91 (40) 2319-4700 | |||
 | synfine@synfinechem.com info@synfinechem.com | |||
| Chemical manufacturer since 2003 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | Ethyl 4-bromophenylacetate |
| Synonyms | 4-Bromophenylacetic acid ethyl ester |
| Molecular Structure | 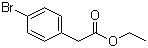 |
| Molecular Formula | C10H11BrO2 |
| Molecular Weight | 243.10 |
| CAS Registry Number | 14062-25-0 |
| EC Number | 628-950-7 |
| SMILES | CCOC(=O)CC1=CC=C(C=C1)Br |
| Melting Point | 29-31 ° |
|---|---|
| Flash point | >110 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Ethyl 4-bromophenylacetate, an organic compound with the formula C10H11BrO2, was first synthesized in the early 20th century as part of research into brominated aromatic esters. This compound is typically produced through the esterification of 4-bromophenylacetic acid with ethanol, using an acid catalyst. The discovery of ethyl 4-bromophenylacetate provided chemists with a useful intermediate for various organic synthesis applications. Ethyl 4-bromophenylacetate is widely used as an intermediate in organic synthesis. Its structure, featuring both an ester and a bromine-substituted aromatic ring, makes it highly reactive and suitable for various chemical transformations. It can undergo nucleophilic substitution reactions, where the bromine atom is replaced by different nucleophiles, leading to the synthesis of diverse compounds. Additionally, the ester group can be hydrolyzed to yield 4-bromophenylacetic acid, which can then be further modified. In the pharmaceutical industry, ethyl 4-bromophenylacetate is used to synthesize active pharmaceutical ingredients (APIs) and intermediates. Its reactivity allows for the introduction of various functional groups necessary for drug development. For instance, it can be used to create compounds with potential anti-inflammatory, analgesic, or anticancer properties. Ethyl 4-bromophenylacetate is employed in the agrochemical sector to produce herbicides, insecticides, and fungicides. The brominated aromatic ester structure allows for the development of agrochemicals with specific biological activities, targeting particular pests or pathogens while minimizing harm to the crops. In materials science, ethyl 4-bromophenylacetate is used in the synthesis of polymers and advanced materials. Its reactivity with various monomers can create polymers with unique properties, such as enhanced durability, chemical resistance, and mechanical strength. These materials are used in coatings, adhesives, and high-performance engineering plastics. Ethyl 4-bromophenylacetate is utilized in academic and industrial research to explore new synthetic methodologies and reaction mechanisms. Researchers use it as a model compound to study substitution reactions, esterification, and other organic processes. In the flavor and fragrance industry, ethyl 4-bromophenylacetate is used as an intermediate to synthesize aroma compounds. Its derivatives can impart pleasant floral and fruity notes, enhancing the sensory qualities of various products, including perfumes, food additives, and personal care items. References 2018. On-demand synthesis of organozinc halides under continuous flow conditions. Nature Protocols. Mateo Berton, Lena Huck, Jesús Alcázar. DOI: 10.1038/nprot.2017.141 2018. C—H Arylation Reactions Using Aryl Halides and Consecutive Photoinduced Electron Transfer (conPET) Catalysts. Science of Synthesis. DOI: SD-229-00253 2015. Synthesis of Esters. Science of Synthesis. DOI: SD-218-00126 |
| Market Analysis Reports |