Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ce Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cepharm.cn | |||
 | +86 (21) 2097-0199 | |||
 | binbin.lin@cepharm.cn | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hebei Medipharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbmedipharm.com | |||
 | +86 (311) 8613-6565 8603-2913 | |||
 | +86 (311) 8696-8724 / 8698-5695 | |||
 | sales@hbmedipharm.com | |||
| Chemical manufacturer since 2004 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Taixing Xingyuan Petrochemical Factory | China | |||
|---|---|---|---|---|
 | www.xingyuanchem.com | |||
 | +86 (438) 622-3708 | |||
 | +86 (438) 622-3000 | |||
 | txzx@hi2000.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Methyl ester compound |
|---|---|
| Name | Methyl 4-hydroxyphenylacetate |
| Synonyms | 4-Hydroxyphenylacetic acid methyl ester |
| Molecular Structure | 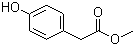 |
| Molecular Formula | C9H10O3 |
| Molecular Weight | 166.18 |
| CAS Registry Number | 14199-15-6 |
| EC Number | 238-050-2 |
| SMILES | COC(=O)CC1=CC=C(C=C1)O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 55-58 °C (Expl.) |
| Index of Refraction | 1.539, Calc.* |
| Boiling Point | 318.8±0.0 °C (760 mmHg), Calc.*, 162-163 °C (5 mmHg) (Expl.) |
| Flash Point | 118.1±13.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Methyl 4-hydroxyphenylacetate is an organic compound with significant importance in pharmaceutical and industrial chemistry. Structurally, it features a phenolic hydroxyl group and an ester group derived from phenylacetic acid. These functional groups impart versatility, making it a valuable intermediate in synthesizing various biologically active compounds and specialty chemicals. The discovery of methyl 4-hydroxyphenylacetate is rooted in the exploration of phenolic esters, known for their natural occurrence and synthetic utility. It is commonly synthesized via esterification of 4-hydroxyphenylacetic acid using methanol and acidic or enzymatic catalysts. Alternative methods include transesterification reactions, offering routes with enhanced selectivity and eco-friendliness. Methyl 4-hydroxyphenylacetate is widely used in the pharmaceutical industry as a building block for drugs. Its phenolic hydroxyl group allows for chemical modifications, such as etherification and acylation, facilitating the synthesis of analgesics, anti-inflammatory agents, and central nervous system drugs. Researchers have explored its role in creating derivatives with improved pharmacokinetics and targeted biological activity. In the cosmetics and personal care industry, methyl 4-hydroxyphenylacetate serves as a precursor for fragrances and flavoring agents. Its ester functionality contributes to its pleasant scent profile, often resembling floral or fruity notes. This property makes it desirable for formulating perfumes, lotions, and other aromatic products. The compound also finds applications in materials science. It is used in polymer chemistry to produce phenolic resins and functionalized polymers, enhancing mechanical properties and thermal stability. Furthermore, its reactivity enables its inclusion in organic electronics and coatings, contributing to advancements in sustainable materials. Ongoing research on methyl 4-hydroxyphenylacetate focuses on developing green synthesis routes and expanding its applications. Enzyme-catalyzed processes have gained attention due to their environmental benefits and high selectivity. Additionally, its derivatives are being explored for potential use in drug discovery and renewable materials. References 2024. Chemical Constituents from the Seeds of Sinapis alba. Chemistry of Natural Compounds, 60(4). DOI: 10.1007/s10600-024-04434-z 2024. Chemical Constituents of Rhinocladiella similis. Chemistry of Natural Compounds, 60(4). DOI: 10.1007/s10600-024-04432-1 2014. Chemical Constituents from the Fruits of Forsythia suspensa and Their Antimicrobial Activity. BioMed Research International. DOI: 10.1155/2014/304830 |
| Market Analysis Reports |