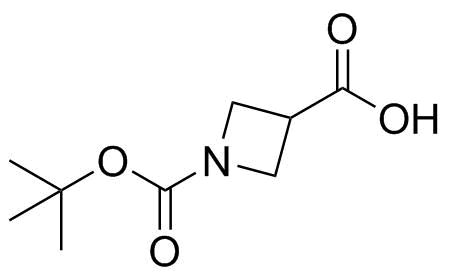
1-N-Boc-3-Azetidinecarboxylic acid is a protected derivative of azetidine-3-carboxylic acid, in which the nitrogen atom of the four-membered azetidine ring is substituted with a tert-butoxycarbonyl (Boc) protecting group. Its structure consists of a strained saturated heterocycle containing one nitrogen atom, a carboxylic acid group at the 3-position, and a Boc-protected secondary amine at the 1-position. The compound belongs to the broader class of constrained amino acid derivatives, which are widely used in medicinal chemistry and peptide design.
The azetidine ring system is a four-membered saturated heterocycle containing one nitrogen atom. Because of its small ring size, azetidine is more strained than five- or six-membered nitrogen heterocycles such as pyrrolidine or piperidine. This ring strain influences both its chemical reactivity and its conformational properties. Substitution at the 3-position with a carboxylic acid introduces a functional handle that allows the compound to be incorporated into more complex molecular frameworks, particularly in peptide-like structures or heterocyclic scaffolds.
The Boc protecting group is a widely used protecting group in organic synthesis for amines. It is introduced to mask the basicity and reactivity of the nitrogen atom during multistep synthesis. In 1-N-Boc-3-azetidinecarboxylic acid, the Boc group stabilizes the azetidine nitrogen, preventing undesired side reactions such as acylation or alkylation. The protecting group is typically stable under neutral and basic conditions but can be removed under acidic conditions, allowing controlled deprotection when needed.
The development of Boc-protected azetidine derivatives is closely related to advances in amino acid and heterocycle chemistry in the late 20th century. As medicinal chemistry increasingly explored conformationally restricted amino acids, azetidine-based scaffolds gained interest due to their ability to impose rigid structural constraints on peptides and small molecules. These constraints can influence biological activity by limiting conformational flexibility and improving binding specificity to biological targets.
Synthesis of 1-N-Boc-3-azetidinecarboxylic acid generally involves construction of the azetidine ring followed by selective functionalization and protection. One common approach starts from suitable amino alcohol or amino acid precursors, followed by cyclization to form the four-membered ring. The nitrogen atom is then protected using di-tert-butyl dicarbonate (Boc2O) under basic conditions to yield the Boc-protected intermediate. The carboxylic acid functionality is introduced or preserved depending on the synthetic route, often through oxidation or substitution reactions on pre-functionalized precursors.
This compound is primarily used as an intermediate in organic synthesis and pharmaceutical research. The combination of a protected amine and a carboxylic acid makes it a useful bifunctional building block for peptide coupling reactions. It can be incorporated into peptide chains or heterocyclic frameworks using standard amide bond-forming techniques. The azetidine ring introduces conformational rigidity, which can improve metabolic stability and receptor selectivity in drug candidates.
In medicinal chemistry, azetidine-containing amino acid derivatives have been explored for their ability to mimic or modify natural amino acids in biologically active molecules. The incorporation of azetidine units can alter the spatial arrangement of functional groups in peptides, potentially leading to improved binding interactions with enzymes or receptors. The Boc-protected form is particularly useful during synthesis because it allows selective manipulation of other functional groups without interference from the nitrogen atom.
In addition to pharmaceutical applications, azetidine derivatives are of interest in materials science and synthetic methodology development. Their strained ring systems can participate in ring-opening reactions under certain conditions, providing access to novel molecular architectures. However, in the Boc-protected form, the nitrogen reactivity is suppressed, allowing controlled use as a stable intermediate.
Overall, 1-N-Boc-3-azetidinecarboxylic acid is a strategically important building block in organic synthesis. Its structure combines a strained azetidine ring with protected amine and carboxylic acid functionality, making it valuable for constructing constrained amino acid analogues and complex molecular frameworks. Its development reflects broader advances in the use of protecting groups and ring-strained heterocycles in modern medicinal and synthetic chemistry.
References
2022. Discovery of small molecule inhibitors of Plasmodium falciparum apicoplast DNA polymerase. Journal of Enzyme Inhibition and Medicinal Chemistry.
DOI: 10.1080/14756366.2022.2070909
2014. A High-Throughput Assay to Identify Inhibitors of the Apicoplast DNA Polymerase from Plasmodium falciparum. Journal of biomolecular screening.
DOI: 10.1177/1087057114528738
|
























































































 GHS07 Warning Details
GHS07 Warning Details