Online Database of Chemicals from Around the World
| Cactus Botanics (Shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cactusbotanics.com | |||
 | +86 (21) 5169-6902 | |||
 | +86 (21) 5106-2932 | |||
 | info@cactusbotanics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Praevideo LLC | Hungary | |||
|---|---|---|---|---|
 | www.praevideo.hu | |||
 | +36 (1) 347-0332 | |||
 | +36 (1) 357-1692 | |||
 | info@praevideo.hu | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Xi'an Union Pharmpro Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.unipharmpro.com | |||
 | +86 (29) 8886-5676 | |||
 | +86 (29) 8886-3915 | |||
 | info@unipharmpro.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shanghai Huich Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.e-biochem.com | |||
 | +86 (21) 6049-6554 | |||
 | 17715331663@163.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Chengdu Push Bio-technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinophytochem.com | |||
 | +86 (028) 8537-0506-229 | |||
 | 3004654993@qq.com | |||
 | QQ Chat | |||
 | WeChat: 18080489829 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Zolanbio Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zolanbio.com | |||
 | +86 13071211027 | |||
 | helen@zolanbio.cn | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Beijing Four Principles Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fourprinciple.cn | |||
 | +86 (10) 8494-7198 | |||
 | +86 (10) 8494-7198 | |||
 | fourprinciple@gmail.com | |||
| Chemical manufacturer since 2004 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Tianjin Jianfeng Natural Product R & D Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jf-natural.com | |||
 | +86 (22) 6623-7098 6623-7213 | |||
 | +86 (22) 6623-7093 | |||
 | sales@jf-natural.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | Zeaxanthin |
| Synonyms | (3R,3'R)-beta,beta-Carotene-3,3'-diol; 4-[18-(4-Hydroxy-2,6,6-trimethyl-1-cyclohexenyl)-3,7,12,16-tetramethyl-octadeca-1,3,5,7,9,11,13,15,17-nonaenyl]-3,5,5-trimethyl-cyclohex-3-en-1-ol |
| Molecular Structure | 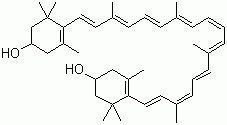 |
| Molecular Formula | C40H56O2 |
| Molecular Weight | 568.88 |
| CAS Registry Number | 144-68-3 |
| EC Number | 205-636-4 |
| SMILES | CC1=C(C(C[C@@H](C1)O)(C)C)/C=C/C(=C/C=C/C(=C/C=C/C=C(/C=C/C=C(/C=C/C2=C(C[C@H](CC2(C)C)O)C)C)C)/C)/C |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 215.5 °C (Expl.) |
| Boiling point | 711.4±55.0 °C 760 mmHg (Calc.)* |
| Flash point | 273.4±26.1 °C (Calc.)* |
| Index of refraction | 1.585 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details | ||||||||
| Safety Statements | P261-P280-P305+P351+P338 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Zeaxanthin is one of the major xanthophyll carotenoids naturally present in plants and certain microorganisms. It was first isolated in the 19th century from corn (Zea mays), from which its name is derived. Zeaxanthin occurs widely in nature together with lutein and other carotenoids in green vegetables, fruits, and flowers, as well as in some algae and cyanobacteria. In higher plants, it is a key component of the xanthophyll cycle, where it functions as part of the protective mechanisms that dissipate excess light energy absorbed during photosynthesis, thereby preventing photooxidative damage to the chloroplast membranes. The biosynthesis of zeaxanthin in plants proceeds through the carotenoid pathway. It begins with the condensation of isopentenyl pyrophosphate units, forming geranylgeranyl pyrophosphate, which then leads to lycopene. Through the sequential action of β-carotene hydroxylase enzymes, lycopene is converted first to β-carotene and subsequently to zeaxanthin. The molecule consists of 40 carbon atoms arranged in a conjugated polyene chain terminated by two β-ionone rings, each containing a hydroxyl group. This structure confers strong antioxidant activity and the ability to interact with biological membranes. In plants, zeaxanthin plays a crucial role in photoprotection through the xanthophyll cycle. Under high light conditions, violaxanthin is de-epoxidized by violaxanthin de-epoxidase to form antheraxanthin and zeaxanthin. When light intensity decreases, zeaxanthin is reconverted to violaxanthin by zeaxanthin epoxidase. This reversible cycle helps regulate non-photochemical quenching of chlorophyll fluorescence, preventing the formation of reactive oxygen species. In animals, zeaxanthin is not synthesized endogenously and must be obtained from dietary sources such as corn, orange peppers, spinach, and eggs. In humans, it accumulates in the retina, particularly in the macular region, where it contributes to visual performance and protection against blue light-induced oxidative stress. Together with lutein, zeaxanthin forms the macular pigment that filters short-wavelength light and reduces chromatic aberration. Epidemiological and clinical studies have linked higher dietary intake of zeaxanthin with a reduced risk of age-related macular degeneration (AMD), a major cause of vision loss in the elderly. Zeaxanthin has also been explored for its potential health benefits beyond ocular protection. Its antioxidant properties are associated with protection against lipid peroxidation and oxidative damage in other tissues. Several clinical and nutritional studies have evaluated zeaxanthin supplementation, often in combination with lutein, showing possible benefits for cognitive health and skin photoprotection. Industrially, zeaxanthin is used as a food colorant and dietary supplement. It can be produced by extraction from natural sources such as paprika and marigold petals or by microbial fermentation using engineered strains of *Paracoccus* or *Flavobacterium* species. Analytical determination of zeaxanthin in biological and food samples is typically performed by high-performance liquid chromatography (HPLC) with photodiode array or mass spectrometric detection. Structural elucidation relies on ultraviolet-visible (UV-Vis) and nuclear magnetic resonance (NMR) spectroscopy, which confirm the trans- and cis-isomeric forms. Research continues to refine biotechnological methods for zeaxanthin production and to understand its physiological roles in plants and humans. Current studies explore its regulation within the carotenoid biosynthetic pathway and its integration into light-harvesting complexes. These efforts contribute to both agricultural improvement and human health applications. References Krinsky NI, Landrum JT & Bone RA (2003) Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annual Review of Nutrition 23 171–201. DOI: 10.1146/annurev.nutr.23.011702.073307 Demmig-Adams B, Gilmore AM & Adams WW III (1996) Carotenoids 3: in vivo function of carotenoids in higher plants. FASEB Journal 10(4) 403–412. DOI: 10.1096/fasebj.10.4.8647339 Mares JA (2016) Lutein and zeaxanthin isomers in eye health and disease. Annual Review of Nutrition 36 571–602. DOI: 10.1146/annurev-nutr-071715-051110 |
| Market Analysis Reports |