Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Yanuo Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yanuo.com | |||
 | +86 (311) 8363-7769 8363-7766 8365-9619 | |||
 | +86 (311) 8362-0118 / 8365-9620 | |||
 | info@yanuo.com | |||
| Chemical manufacturer since 1994 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Weifang Sunwin Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunwincn.com | |||
 | +86 (536) 886-9166 | |||
 | +86 (536) 886-9188 | |||
 | sales@sunwincn.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Novasyn Organics Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.novasynorganics.com | |||
 | +91 (406) 620-2233 633-9111 | |||
 | +91 (402) 781-1997 | |||
 | info@novasynorganics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yuchuang Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuchuang-chem.com | |||
 | +86 (21) 6143-1639 +86 13817846823 | |||
 | +86 (21) 6143-1639 | |||
 | sales@yuchuang-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Cyanochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cyanochem.com | |||
 | +86 (571) 8522-0831 +86 17788583750 | |||
 | +86 (571) 8522-0831 | |||
 | info@cyanochem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shijiazhuang C-element Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.c-elementbiotech.com | |||
 | +86 18033710157 | |||
 | Lisa@C-elementBiotech.com | |||
 | QQ Chat | |||
 | WeChat: 18033710157 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| Chem Service, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemservice.com | |||
 | +1 (610) 692-3026 | |||
 | +1 (610) 692-8729 | |||
 | info@chemservice.com | |||
| Chemical manufacturer since 1962 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| True Pharmachem Inc. | USA | |||
|---|---|---|---|---|
 | www.truepharmachem.com | |||
 | +1 (217) 766-6932 | |||
 | +1 (973) 588-3470 | |||
 | sales@truepharmachem.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Aminopyridine |
|---|---|
| Name | 4-Amino-2-chloropyridine |
| Synonyms | 2-Chloro-4-pyridinamine |
| Molecular Structure | 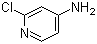 |
| Molecular Formula | C5H5ClN2 |
| Molecular Weight | 128.56 |
| CAS Registry Number | 14432-12-3 |
| EC Number | 238-403-0 |
| SMILES | C1=CN=C(C=C1N)Cl |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 90-94 °C (Expl.) |
| Index of Refraction | 1.607, Calc.* |
| Boiling Point | 299.9±20.0 °C (760 mmHg), Calc.*, 350.6 °C (Expl.) |
| Flash Point | 135.2±21.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
4-Amino-2-chloropyridine is an aromatic heterocyclic compound with the molecular formula C5H5ClN2. It consists of a pyridine ring substituted at the 2-position with a chlorine atom and at the 4-position with an amino group. This compound belongs to the class of chloropyridine derivatives, which are frequently studied due to their chemical reactivity and utility in pharmaceutical and agrochemical synthesis. The synthesis of 4-amino-2-chloropyridine has been documented through several reliable and reproducible methods. One of the most established routes involves the nucleophilic substitution of 2,4-dichloropyridine, where the chlorine atom at the 4-position is replaced by an amino group using ammonia or an amine source under appropriate conditions. This selective substitution exploits the differential reactivity of halogenated positions on the pyridine ring. The reaction conditions are typically mild, and the product is obtained with good yields and purity after purification by recrystallization or chromatographic methods. 4-Amino-2-chloropyridine is a valuable intermediate in the synthesis of more complex nitrogen-containing compounds. The presence of both an electron-donating amino group and an electron-withdrawing chlorine atom on the pyridine ring enhances its reactivity in various transformations, including cross-coupling reactions and heterocycle formation. The amino group serves as a nucleophilic center, enabling acylation, sulfonation, and diazotization reactions. Meanwhile, the chlorine atom at the 2-position can undergo substitution via palladium-catalyzed cross-coupling methods such as Suzuki, Heck, or Buchwald-Hartwig reactions, allowing for the formation of C–C or C–N bonds. In medicinal chemistry, 4-amino-2-chloropyridine is widely used as a building block in the development of bioactive compounds. It is employed in the synthesis of various pharmaceutical candidates, particularly those targeting kinase enzymes, bacterial infections, and inflammatory pathways. The compound’s pyridine nucleus is known to exhibit favorable pharmacokinetic properties, and its substitution pattern allows for the generation of derivatives with high binding specificity and metabolic stability. Several derivatives of 4-amino-2-chloropyridine have been reported in the literature with demonstrated biological activities. For example, it has been used as a precursor for the preparation of inhibitors targeting protein tyrosine kinases and cyclin-dependent kinases. It also appears in synthetic routes leading to pyridine-based ureas, amides, and sulfonamides, which are known classes of compounds with antibacterial, antiviral, and anticancer applications. While the parent compound itself is not typically used as a drug, its role in facilitating access to bioactive molecules is well recognized. In the field of agrochemical research, 4-amino-2-chloropyridine has found use in the synthesis of herbicidal and fungicidal agents. Pyridine derivatives are frequently incorporated into agrochemical structures due to their ability to interfere with plant or microbial biochemical processes. The structural framework of 4-amino-2-chloropyridine allows for chemical diversification, which supports the discovery of compounds with selective activity against agricultural pests or pathogens. Analytical characterization of 4-amino-2-chloropyridine is routinely conducted using techniques such as nuclear magnetic resonance spectroscopy, mass spectrometry, and infrared spectroscopy. In NMR spectra, signals corresponding to the pyridine ring protons, as well as the amino group, provide clear evidence of the compound’s structure. The infrared spectrum typically shows characteristic bands for N–H stretching and C–Cl vibrations. The compound is generally isolated as a crystalline solid, with good stability under ambient storage conditions, and is soluble in polar organic solvents such as ethanol, methanol, and dimethylformamide. The compound should be handled with care, as is standard for halogenated and aminated aromatic substances. Appropriate personal protective equipment and ventilation are required during handling and synthesis. 4-Amino-2-chloropyridine continues to serve as a versatile and well-studied intermediate in organic and medicinal chemistry, with a clearly documented history of applications across both industrial and academic research settings. References 2024. Physical and Chemical Characterisations, Optical Properties and Dielectric Studies of a New Organic-Inorganic Material: bis(4-amino-2-chloropyridinium) Tetrachloromercurate (II) Monohydrate. Chemistry Africa, 7(3). DOI: 10.1007/s42250-023-00813-1 2023. Exploiting Ion-Dipole and Ion-Exchange Interactions for the Removal of Aminopyridines from Aqueous Environments Using Polymer Inclusion Membranes. Chemistry Africa, 6(5). DOI: 10.1007/s42250-023-00734-z 2018. Preparation, molecular structure, thermal properties, electrical conductivity analysis and dielectric relaxation of a new hybrid compound (NH2C5H3ClNH)2ZnBr4·H2O. Chemical Papers, 72(12). DOI: 10.1007/s11696-018-0521-8 |
| Market Analysis Reports |