Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Caerulum Pharma Discovery Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.caerulumpharm.com | |||
 | +86 (21) 6455-6180 +86 189149758185 | |||
 | +86 (21) 6455-6180 | |||
 | sales-cpd@caerulumpharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Name | (S,R,S)-AHPC |
|---|---|
| Synonyms | (2,5-dioxopyrrolidin-1-yl) 6-[(2Z)-1,1-dimethyl-2-[(2E,4E)-5-(1,1,3-trimethylbenzo[e]indol-3-ium-2-yl)penta-2,4-dienylidene]benzo[e]indol-3-yl]hexanoate;chloride |
| Molecular Structure | 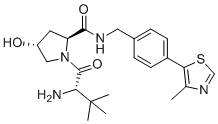 |
| Molecular Formula | C22H30N4O3S |
| Molecular Weight | 430.56 |
| Protein Sequence | XX |
| CAS Registry Number | 1448297-52-6 |
| EC Number | 894-674-0 |
| SMILES | CC1=C(SC=N1)C2=CC=C(C=C2)CNC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)N)O |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.604, Calc.* |
| Boiling Point | 691.6±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 372.1±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
(S,R,S)-AHPC, or (S,R,S)-2-(2-amino-3-hydroxypropyl)-4-(4-(2-oxopyrrolidin-1-yl)phenyl)-2,4-dihydro-3H-1,2,4-triazole-3-thione, is a novel compound of great interest in drug development and chemical biology. (S,R,S)-AHPC has a complex structure and potential therapeutic applications. The discovery of (S,R,S)-AHPC stems from the growing interest in proteolysis targeting chimeras (PROTACs), which are used to degrade specific proteins implicated in disease. Researchers developed (S,R,S)-AHPC to exploit this approach by combining it with small molecule ligands to create bifunctional molecules capable of inducing selective protein degradation to overcome resistance mechanisms in the treatment of cancer and other diseases. Its triazole-thione core is responsible for molecular stability and constitutes the central scaffold for interaction with target proteins; the amino-hydroxypropyl group enhances solubility and promotes interaction with biological targets; and the oxopyrrolidino-phenyl group contributes to the binding specificity and biological activity of the compound.  (S,R,S)-AHPC is used to design PROTACs, which are bifunctional molecules that recruit E3 ubiquitin ligases to target proteins, marking them for proteasomal degradation. This strategy allows for the selective degradation of pathogenic proteins, providing potential treatments for diseases such as cancer, neurodegenerative diseases, and inflammatory diseases. In cancer research, (S,R,S)-AHPC-based PROTACs target oncogenic proteins that drive tumor growth and survival. By degrading these proteins, these compounds can inhibit cancer cell proliferation and induce apoptosis, providing a new mechanism for combating drug-resistant cancers. (S,R,S)-AHPC is used as a chemical probe to study protein-protein interactions and cellular pathways. Its ability to induce targeted protein degradation helps to elucidate the function and role of specific proteins in biological systems, thereby advancing the understanding of cellular mechanisms and disease pathology. (S,R,S)-AHPC facilitates functional studies of proteins by achieving conditional knockdown of targeted proteins. This approach helps to dissect the biological role of proteins in various cellular contexts and facilitates the discovery of new drug targets and therapeutic strategies. (S,R,S)-AHPC serves as a lead compound in drug programs to develop targeted therapeutics for diseases with unmet medical needs. Its unique properties make it a valuable starting point for the design and optimization of novel therapeutics that exploit protein degradation mechanisms. (S,R,S)-AHPC is also used in SAR studies to optimize the efficacy, selectivity, and pharmacokinetic properties of (S,R,S)-AHPC derivatives. These studies provide information for the design of next-generation PROTACs and other therapeutic molecules. References 2022-02-04. Discovery of Potent, Selective, and In Vivo Efficacious AKT Kinase Protein Degraders via Structure-Activity Relationship Studies DOI: 10.1021/acs.jmedchem.1c02165 2021-12-02. Design, Synthesis, and Evaluation of Potent, Selective, and Bioavailable AKT Kinase Degraders DOI: 10.1021/acs.jmedchem.1c01476 2021-09-16. Identifying transcriptional programs underlying cancer drug response with TraCe-seq DOI: 10.1038/s41587-021-01005-3 |
| Market Analysis Reports |