Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Name | 3-Methyl-1-(6-methyl-1H-benzimidazol-2-yl)-4-(phenylmethyl)-1H-pyrazol-5-ol |
|---|---|
| Synonyms | HUHS 015 |
| Molecular Structure | 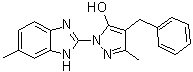 |
| Molecular Formula | C19H18N4O |
| Molecular Weight | 318.37 |
| CAS Registry Number | 1453097-13-6 |
| SMILES | CC1=CC2=C(C=C1)N=C(N2)N3C(=O)C(=C(N3)C)CC4=CC=CC=C4 |
| Solubility | Insoluble (8.6E-3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.30±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2016 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
3-Methyl-1-(6-methyl-1H-benzimidazol-2-yl)-4-(phenylmethyl)-1H-pyrazol-5-ol, commonly known as MMMP, is a heterocyclic ring Compound, belonging to benzimidazole derivatives. MMMP is considered a promising scaffold with bioactive and pharmacological properties that warrants further investigation. MMMP exhibits potent antimicrobial activity against a variety of pathogenic microorganisms, including bacteria, fungi, and protozoa, making it a potential candidate for pharmaceuticals, personal care products, and agricultural applications. MMMP has significant antioxidant properties, capable of scavenging free radicals and preventing oxidative damage to cells and tissues. MMMP exerts anti-inflammatory effects by modulating inflammatory pathways and inhibiting the release of pro-inflammatory mediators such as cytokines and chemokines. MMMP has shown promising anticancer potential in preclinical studies through its ability to inhibit cancer cell proliferation, induce apoptosis, and inhibit tumor growth. MMMPs exert neuroprotective effects by attenuating neuronal damage, reducing oxidative stress, and promoting neuronal survival in various neurodegenerative and neurological disorders. Its ability to cross the blood-brain barrier and modulate neuronal function makes it a promising candidate for treating diseases such as Alzheimer's disease, Parkinson's disease and ischemic stroke. References 2015. Improving the bioavailability and anticancer effect of the PCA-1/ALKBH3 inhibitor HUHS015 using sodium salt. In vivo (Athens, Greece), 29, 1. URL: https://pubmed.ncbi.nlm.nih.gov/25600528 2014. Design and synthesis of prostate cancer antigen-1 (PCA-1/ALKBH3) inhibitors as anti-prostate cancer drugs. Bioorganic & Medicinal Chemistry Letters, 24, 4. DOI: 10.1016/j.bmcl.2014.01.008 2002. Effect of loading rate on the apparent fracture toughness of acrylic bone cement. Bio-medical materials and engineering, 12, 3. URL: https://pubmed.ncbi.nlm.nih.gov/12122238 |
| Market Analysis Reports |