Online Database of Chemicals from Around the World
| Shenyang OllyChem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ollychem.com | |||
 | +86 (24) 6225-9849 +86 13840042106 | |||
 | +86 (24) 6225-9831 | |||
 | info@ollychem.com oliverdu@ollychem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Forever Synthesis Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.foreversyn.com | |||
 | +86 (551) 6288-8437 +86 18096409024 | |||
 | sales@foreversyn.com sales02@foreversyn.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | 15(S)-Latanoprost |
| Synonyms | 15-epi Latanoprost; (Z)-7-[(1R,2R,3R,5S)-3,5-Dihydroxy-2-[(3R)-3-hydroxy-5-phenylpentyl]cyclopentyl]hept-5-enoic acid isopropyl ester |
| Molecular Structure | 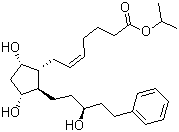 |
| Molecular Formula | C26H40O5 |
| Molecular Weight | 432.59 |
| CAS Registry Number | 145773-22-4 |
| EC Number | 687-773-3 |
| SMILES | CC(C)OC(=O)CCC/C=CC[C@H]1[C@H](C[C@H]([C@@H]1CC[C@@H](CCC2=CC=CC=C2)O)O)O |
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H361 Details | ||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P316-P318-P321-P330-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
15(S)-Latanoprost is a stereoisomer of the widely used pharmaceutical latanoprost, which belongs to the class of prostaglandin analogs. The discovery of 15(S)-Latanoprost was part of research efforts aimed at developing more effective treatments for glaucoma and ocular hypertension. During the synthesis of prostaglandin analogs, stereochemistry plays a critical role in determining the biological activity of the compounds. The 15(S) configuration refers to the specific spatial arrangement of the hydroxyl group on the carbon chain, which is crucial for the molecule’s interaction with prostaglandin receptors in the eye. 15(S)-Latanoprost was identified through stereochemical studies of latanoprost, a drug developed in the 1990s. While the biologically active form of latanoprost is the 15(R) isomer, the 15(S)-stereoisomer has been synthesized and studied to understand the impact of stereochemistry on pharmacological activity. This stereoisomer does not exhibit the same potency in reducing intraocular pressure as the 15(R)-isomer, which is the active ingredient in commercial latanoprost formulations. Nevertheless, its discovery has provided valuable insights into the structure-activity relationships of prostaglandin analogs. The application of 15(S)-Latanoprost is primarily in research rather than clinical use. It serves as a reference compound for scientists studying the effects of stereochemistry on prostaglandin analogs and their interactions with biological systems. By comparing the activity of the 15(S)- and 15(R)-isomers, researchers can gain a better understanding of how molecular configuration influences drug efficacy and receptor binding. While 15(S)-Latanoprost does not have therapeutic applications, its role in scientific studies has been instrumental in refining the design of prostaglandin-based drugs. The information gathered from such research has contributed to the development of newer and more effective treatments for glaucoma and other conditions involving elevated intraocular pressure. References 1993. Phenyl-substituted prostaglandins: potent and selective antiglaucoma agents. Journal of Medicinal Chemistry, 36, 2. URL: https://pubmed.ncbi.nlm.nih.gov/8423596 |
| Market Analysis Reports |