Online Database of Chemicals from Around the World
| Alway Chem, China | China | |||
|---|---|---|---|---|
 | www.alwaychem.net | |||
 | +86 (532) 8586-4000 | |||
 | +86 (532) 8589-3133 | |||
 | alwaychem@gmail.com alwaychem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Qyubiotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qyubiotech.com | |||
 | +86 (021) 3759-6619 | |||
 | +86 (021) 3759-6619 | |||
 | lioner@qyubiotech.com | |||
 | QQ Chat | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Fujian Wolfa Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wolfabio.com | |||
 | +86 18050950397 | |||
 | jomin@wolfabio.com | |||
 | WhatsApp:+86 18359103607 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty alcohol |
|---|---|
| Name | 1-Methylcyclopentanol |
| Molecular Structure | 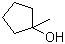 |
| Molecular Formula | C6H12O |
| Molecular Weight | 100.16 |
| CAS Registry Number | 1462-03-9 |
| EC Number | 215-963-4 |
| SMILES | CC1(CCCC1)O |
| Density | 0.904 |
|---|---|
| Melting point | 31-36 °C |
| Boiling point | 135-136 °C |
| Flash point | 40 °C |
| Hazard Symbols |    GHS02;GHS05;GHS07 Danger Details GHS02;GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H228-H302-H315-H318-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P240-P241-P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P305+P354+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1325 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1-Methylcyclopentanol is an organic compound with the formula C6H11OH. It is a colorless liquid with a characteristic odor, commonly used in various industrial and chemical applications due to its versatile chemical properties. The discovery of 1-methylcyclopentanol can be traced back to the early 20th century when researchers were exploring the properties and reactions of cyclopentanol derivatives. Its synthesis typically involves the alkylation of cyclopentanol with methyl iodide in the presence of a base. This reaction introduces the methyl group into the cyclopentanol structure, resulting in 1-methylcyclopentanol. In industrial applications, 1-methylcyclopentanol is primarily used as an intermediate in the synthesis of other chemicals. It serves as a valuable building block for the production of various pharmaceuticals, agrochemicals, and specialty chemicals. Its unique structure allows it to participate in a range of chemical reactions, making it a useful compound in synthetic chemistry. One notable application of 1-methylcyclopentanol is in the production of fragrance compounds. Its pleasant odor makes it suitable for use in perfumes and other aromatic products. Additionally, it is employed in the formulation of certain plasticizers and resins, where its properties contribute to the flexibility and durability of the final products. Another significant application of 1-methylcyclopentanol is in the research and development of new chemical processes. Its reactivity and structure make it a useful substrate for studying various chemical transformations and mechanisms. Researchers use it to investigate reaction pathways and develop new synthetic routes for producing complex molecules. Overall, 1-methylcyclopentanol is an important compound in the field of organic chemistry and industrial applications. Its discovery and subsequent utilization in various chemical processes highlight its value as an intermediate and its contributions to the development of new materials and products. References 2013. Catalysts for hydration of cyclohexene to cyclohexanol. Chemical Research in Chinese Universities, 29, 4. DOI: 10.1007/s40242-013-3091-x 2004. From Alkyl Halides Using Activated Rieke Copper. Science of Synthesis, 3. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-003-00362 2004. beta-Heteroalkylmercury(II) Compounds from Oxymercuration of Alkenes. Science of Synthesis, 3. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-003-00250 |
| Market Analysis Reports |