Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | API >> Antiparasitic drug >> Antimalarial |
|---|---|
| Name | 8-Hydroxyquinoline |
| Synonyms | 8-Quinolinol; Quinolin-8-ol; Oxine |
| Molecular Structure | 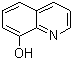 |
| Molecular Formula | C9H7NO |
| Molecular Weight | 145.16 |
| CAS Registry Number | 148-24-3 |
| EC Number | 205-711-1 |
| SMILES | C1=CC2=C(C(=C1)O)N=CC=C2 |
| Solubility | 10mM (DMSO) |
|---|---|
| Melting point | 70-74 °C |
| Boiling point | 267 °C (752 mmHg) |
| Hazard Symbols |      GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details GHS05;GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H317-H318-H360-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P270-P272-P273-P280-P301+P316-P301+P317-P302+P352-P305+P354+P338-P317-P318-P321-P330-P333+P317-P362+P364-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
8-Hydroxyquinoline, also known as oxine, was first synthesized in the late 19th century by the German chemist Johannes Thiele in 1899. This heterocyclic organic compound is derived from quinoline, a structure featuring a nitrogen atom within a benzene ring, where the 8-hydroxy position signifies the attachment of a hydroxyl group at the eighth position. The significance of 8-Hydroxyquinoline grew with the recognition of its chelating properties, forming stable complexes with metal ions. This ability has rendered it a valuable reagent in both industrial and scientific applications. One of the primary uses of 8-Hydroxyquinoline is in analytical chemistry for the detection and separation of metals. It forms stable, often fluorescent, complexes with metals such as aluminum, zinc, and copper. These chelating properties facilitate its use in metallurgy, water purification, and soil remediation by removing heavy metal contaminants. 8-Hydroxyquinoline and its derivatives exhibit potent antimicrobial activity against bacteria, fungi, and protozoa. This has led to its incorporation in antiseptic creams, disinfectants, and preservatives. For instance, 8-hydroxyquinoline sulfate is used in medicinal products to treat topical infections and as a preservative in cosmetics and pharmaceuticals to prevent microbial growth. In the realm of biochemistry and molecular biology, 8-Hydroxyquinoline is used as a reagent to inhibit metalloproteinase enzymes, which are critical in studying protein functions and structures. Its ability to chelate essential metal ions makes it a useful tool in enzyme inhibition studies and the exploration of cellular processes dependent on metal ions. The agricultural sector benefits from 8-Hydroxyquinoline's fungicidal properties. It is used as a fungicide to protect crops from fungal infections, enhancing yield and quality. Additionally, its role in plant tissue culture as a growth medium supplement helps in the successful propagation of plant cells and tissues. In the field of materials science, 8-Hydroxyquinoline complexes, particularly with aluminum (Alq3), are pivotal in the development of organic light-emitting diodes (OLEDs). These materials are essential in creating modern display technologies used in screens for smartphones, televisions, and other electronic devices. References 2007. Possible involvement of genes on the Q chromosome of Nicotiana tabacum in expression of hybrid lethality and programmed cell death during interspecific hybridization to Nicotiana debneyi. Planta, 226, 1. DOI: 10.1007/s00425-007-0522-2 2006. Mono- and Di(dimethylamino)styryl-Substituted Borondipyrromethene and Borondiindomethene Dyes with Intense Near-Infrared Fluorescence. Chemistry - An Asian Journal, 1, 5. DOI: 10.1002/asia.200600042 1965. Experimental studies on 8-hydroxyquinoline in rats and mice. Archives of pathology, 79, 3. URL: https://pubmed.ncbi.nlm.nih.gov/14246204 |
| Market Analysis Reports |