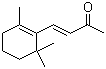
Irisone is a naturally occurring ketone found in the essential oils of iris species, particularly in orris root (Iris germanica, Iris pallida, and Iris florentina). It is classified as an ionone derivative and contributes significantly to the characteristic violet-like fragrance of these plants. The compound was identified through early studies of the volatile components of iris rhizomes, where it was found to be responsible for the distinct and highly valued scent. Advances in chromatographic techniques facilitated the isolation and structural elucidation of irisone, allowing for further research into its properties and applications.
The primary application of irisone is in the fragrance industry, where it is used in the formulation of high-end perfumes and cosmetics. The compound’s floral, woody, and powdery scent makes it a valuable ingredient in luxury fragrance compositions, particularly those designed to mimic the aroma of violets or enhance floral bouquets. Due to the extensive time required for orris root to develop its aromatic compounds, natural extraction remains a costly process. As a result, synthetic irisone has been developed to provide a more accessible and consistent supply for commercial use.
Beyond perfumery, irisone has been incorporated into various personal care and cosmetic products, including lotions, soaps, and powders. Its ability to impart a long-lasting and elegant fragrance has made it a favored ingredient in high-quality formulations. The stability and compatibility of irisone with other fragrance compounds further enhance its utility in these applications.
The compound has also been studied in the context of flavoring, though its use in food and beverages is less prominent than in perfumery. It has been explored as a component in flavor formulations requiring subtle floral and woody notes. The controlled use of irisone in such applications follows industry regulations and safety assessments.
In industrial and material science research, irisone has been examined for its potential interactions with polymers and other substrates in fragrance delivery systems. The controlled release of volatile compounds in textiles, packaging, and other consumer goods is an area of interest, with irisone contributing to long-lasting scent applications.
The discovery and subsequent applications of irisone have solidified its place as a key compound in the fragrance and cosmetics industries. Ongoing research continues to refine its synthesis and optimize its use in commercial formulations while ensuring compliance with safety standards and regulatory guidelines.
References
14901-07-6|Irisone|2024. Combined metabolic engineering and lipid droplets degradation to increase vitamin A production in Saccharomyces cerevisiae. Microbial Cell Factories, 23(1).
DOI: 10.1186/s12934-024-02596-7
2024. DNA binding, and apoptosis-inducing activities of a β-ionone-derived ester in human myeloid leukemia cells: multispectral and molecular dynamic simulation analyses. Scientific Reports, 14(1).
DOI: 10.1038/s41598-024-78690-y
2024. β-ionone inhibits the grazing of Daphnia sinensis by reducing the activity of acetylcholinesterase. Journal of Hazardous Materials, 479.
DOI: 10.1016/j.jhazmat.2024.135690
|




























 GHS09 Warning Details
GHS09 Warning Details