Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| La Mesta Chimie Fine S.A.S. | France | |||
|---|---|---|---|---|
 | www.la-mesta.com | |||
 | +33 (4) 9208-5300 | |||
 | +33 (4) 9208-5340 | |||
 | info@la-mesta.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou TJM Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjmchem.com | |||
 | +86 13388471767 | |||
 | +86 (571) 8822-3279 | |||
 | sales@tjmchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Sai Chemie Inc. | India | |||
|---|---|---|---|---|
 | www.saichemie.com | |||
 | +91 (816) 935-6778 | |||
 | saichemicalinc@gmail.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nivon Specialties | India | |||
|---|---|---|---|---|
 | www.navoneindia.com | |||
 | +91 9323789882 | |||
 | nivonpharma@yahoo.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | 3-Methoxyphenol |
| Synonyms | 3-Hydroxyanisole |
| Molecular Structure | 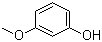 |
| Molecular Formula | C7H8O2 |
| Molecular Weight | 124.14 |
| CAS Registry Number | 150-19-6 |
| EC Number | 205-754-6 |
| SMILES | COC1=CC=CC(=C1)O |
| Density | 1.1±0.1 g/cm3 Calc.*, 1.145 g/mL (Expl.) |
|---|---|
| Melting point | -18 °C (Expl.), -18 °C (Expl.) |
| Boiling point | 244.3 °C 760 mmHg (Calc.)*, 293.6 - 296.5 °C (Expl.) |
| Flash point | 119.1±4.8 °C (Calc.)*, 124 °C (Expl.) |
| Solubility | waterr: slightly soluble (Expl.) |
| Index of refraction | 1.535 (Calc.)*, 1.552 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H315-H318-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
3-Methoxyphenol, also known as m-methoxyphenol or resorcinol monomethyl ether, is an aromatic organic compound with the molecular formula C7H8O2. It is one of the three isomeric forms of methoxyphenol, distinguished by the position of the methoxy group (–OCH3) relative to the hydroxyl group (–OH) on the benzene ring. In 3-methoxyphenol, the methoxy group is located at the meta position (position 3) with respect to the hydroxyl group. This compound appears as a colorless to slightly yellow liquid at room temperature and has a distinctive phenolic odor. It is moderately soluble in water and highly soluble in organic solvents such as ethanol, ether, and chloroform. Its structure imparts both hydrophilic and hydrophobic properties, making it versatile in organic synthesis and formulation chemistry. 3-Methoxyphenol has been studied and applied in a number of industrial and research contexts. It is commonly used as an intermediate in the synthesis of dyes, agrochemicals, and pharmaceuticals. Its phenolic and methoxy substituents allow for functional group transformations and electrophilic substitution reactions that enable the creation of more complex molecules. In the field of medicinal chemistry, derivatives of 3-methoxyphenol have been investigated for their antimicrobial, antioxidant, and anti-inflammatory activities. Historically, 3-methoxyphenol and its isomers have attracted interest as naturally occurring phenolic compounds in plants. Compounds with similar structures are found in essential oils and plant resins, where they contribute to fragrance and bioactivity. Synthetic analogs like 3-methoxyphenol are studied to understand structure–activity relationships and to develop new bioactive molecules for therapeutic and cosmetic use. In cosmetics and personal care products, 3-methoxyphenol can serve as a fragrance ingredient or as a component in formulations with antioxidant functions. However, as with many phenolic compounds, its use is regulated due to potential skin sensitization risks at high concentrations. Toxicological assessments indicate that while 3-methoxyphenol has relatively low acute toxicity, proper handling and formulation limits are required to ensure safety in consumer products. Environmental data suggest that 3-methoxyphenol is biodegradable under aerobic conditions and has low potential for bioaccumulation. It is generally considered to have low environmental persistence when released in small quantities. Nonetheless, as with other aromatic compounds, care must be taken to prevent excessive environmental exposure. In summary, 3-methoxyphenol is a valuable chemical intermediate with applications in organic synthesis, pharmaceuticals, and cosmetics. Its structural features and reactivity make it a useful building block for a variety of chemical transformations, and its moderate toxicity and environmental profile support its use in controlled industrial settings. References 2023. Recent advancement in development and modification of nanofibrous matrix for the application in sensing and remediation of water pollutants. Applied Nanoscience. DOI: 10.1007/s13204-023-02844-z 2023. Carboxylated chalcones and related flavonoids as inhibitors of xanthine oxidase. Medicinal Chemistry Research. DOI: 10.1007/s00044-023-03109-8 2021. Hydroxylation of Arylboronic Acids to Phenols on a Vanadia-Titania Catalyst. Synfacts. DOI: 10.1055/s-0040-1720712 |
| Market Analysis Reports |