Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Targetmol China | China | |||
|---|---|---|---|---|
 | www.targetmol.cn | |||
 | +86 (400) 820-0310 | |||
 | sales@targetmol.cn | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Classification | Flavors and spices >> Synthetic spice >> Alcoholic spice >> Terpene alcohol |
|---|---|
| Name | 6-Hydroxytaxol |
| Synonyms | [(1S,2S,3R,4S,7R,8S,9R,10S,12R,15S)-4,12-diacetyloxy-15-[(2R,3S)-3-benzamido-2-hydroxy-3-phenylpropanoyl]oxy-1,8,9-trihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7]heptadec-13-en-2-yl] benzoate |
| Molecular Structure | 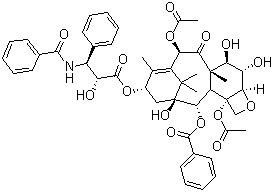 |
| Molecular Formula | C47H51NO15 |
| Molecular Weight | 869.91 |
| CAS Registry Number | 153212-75-0 |
| EC Number | 687-556-3 |
| SMILES | CC1=C2[C@H](C(=O)[C@]3([C@H]([C@@H]([C@@](C2(C)C)(C[C@@H]1OC(=O)[C@@H]([C@H](C4=CC=CC=C4)NC(=O)C5=CC=CC=C5)O)O)OC(=O)C6=CC=CC=C6)[C@@]7(CO[C@@H]7[C@H]([C@@H]3O)O)OC(=O)C)C)OC(=O)C |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 979.8±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 546.4±34.3 °C (Calc.)* |
| Index of refraction | 1.646 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS08 Danger Details GHS05;GHS08 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H318-H340-H360 Details | ||||||||||||||||
| Safety Statements | P203-P264+P265-P280-P305+P354+P338-P317-P318-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
6-Hydroxytaxol is a chemically modified derivative of paclitaxel, a well-known natural product originally isolated from the bark of the Pacific yew tree. Paclitaxel is widely recognized for its potent antitumor activity, primarily functioning by stabilizing microtubules and thereby inhibiting cell division. The introduction of a hydroxyl group at the 6-position of the taxane core structure results in 6-hydroxytaxol, a compound studied for its biological properties and potential applications in cancer treatment research. The discovery of 6-hydroxytaxol arose from efforts to understand structure-activity relationships within the taxane family and to explore derivatives that might improve solubility, efficacy, or pharmacokinetic profiles compared to paclitaxel. Chemical modification at the 6-position was of particular interest due to its location on the taxane ring, which can influence the compound’s interaction with microtubules and its metabolic stability. In vitro studies have demonstrated that 6-hydroxytaxol retains microtubule-stabilizing activity similar to that of paclitaxel, contributing to the disruption of microtubule dynamics essential for mitotic spindle formation during cell division. This activity underlies its cytotoxic effects against various cancer cell lines. Moreover, the hydroxyl substitution can affect the compound’s affinity for tubulin and its susceptibility to metabolic enzymes, factors critical for drug efficacy and duration of action. The application of 6-hydroxytaxol has primarily been within experimental and preclinical research contexts, where it serves as a tool to investigate the effects of structural modifications on taxane pharmacology. Researchers have explored its potential to overcome limitations associated with paclitaxel, such as multidrug resistance and limited solubility, although it has not been developed into a widely used clinical agent. 6-Hydroxytaxol has also been useful in studies of drug metabolism and detoxification, as its hydroxyl group provides a site for conjugation reactions, such as glucuronidation or sulfation, facilitating investigation into how taxane derivatives are processed in the body. Understanding these metabolic pathways is important for optimizing dosing regimens and minimizing adverse effects. In summary, 6-hydroxytaxol represents a chemically modified taxane compound investigated for its microtubule-stabilizing and cytotoxic properties. It is mainly utilized in research to explore taxane structure-activity relationships and metabolism, contributing to the broader knowledge base that supports the development of improved anticancer therapies. References 1994. Isolation, Structural Determination, and Biological Activity of 6.alpha.-Hydroxytaxol, the Principal Human Metabolite of Taxol. Journal of Medicinal Chemistry, 37(5). DOI: 10.1021/jm00031a022 1995. Comparative in vitro cytotoxic effects of taxol and its major human metabolite 6 alpha-hydroxytaxol. Cancer Chemotherapy and Pharmacology, 36(1). DOI: 10.1007/bf00689197 2018. A dried blood spot assay for paclitaxel and its metabolites. Journal of Pharmaceutical and Biomedical Analysis, 149. DOI: 10.1016/j.jpba.2017.10.007 |
| Market Analysis Reports |