Online Database of Chemicals from Around the World
| Vishnupriya Chemicals Private Limited | India | |||
|---|---|---|---|---|
 | vishnupriya.in | |||
 | +91 9676770180 | |||
 | web@vishnupriya.in | |||
| Chemical manufacturer since 1979 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Name | Lead acetate anhydrous |
|---|---|
| Synonyms | Lead(2+) diacetate; Plumbous Diacetate; |
| Molecular Structure | 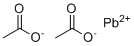 |
| Molecular Formula | (CH3COO)2Pb |
| Molecular Weight | 325.29 |
| CAS Registry Number | 15347-57-6 |
| EC Number | 239-379-4 |
| SMILES | CC(=O)[O-].CC(=O)[O-].[Pb+2] |
| Solubility | 44.3 g/100mL (water 20 °C) |
|---|---|
| Density | 2.55 g/mL |
| Melting point | 280 °C (Epxl.) |
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H332-H360-H373-H400-H410 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P270-P271-P273-P280-P301+P317-P304+P340-P317-P318-P319-P330-P391-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
|
Lead acetate anhydrous, with the chemical formula Pb(C₂H₃O₂)₂, is a white, crystalline compound that has been historically important in various industrial and chemical processes. It is also known by its common name "sugar of lead" due to its sweet taste, which was discovered in antiquity. Lead acetate anhydrous is primarily used in the production of other chemicals, including lead-based compounds, but its use has declined over time due to health concerns related to lead exposure. Despite this, it remains an important chemical in specific industrial applications, particularly in the fields of textiles, leather, and the manufacture of other chemicals. The discovery of lead acetate dates back to ancient times. The compound was likely first produced by the Egyptians, who were known to have extracted and used lead salts. The process of making lead acetate, however, was well documented in Europe during the 17th and 18th centuries. The compound was initially synthesized by reacting lead with acetic acid, a method still used today in modern chemistry. Its sweet taste, which gave rise to the name "sugar of lead," contributed to its early popularity, although its toxic properties were not immediately understood. Lead acetate anhydrous has a range of applications, particularly in the industrial sector. One of its earliest uses was in the textile industry, where it was employed as a mordant in the dyeing of fabrics. The ability of lead acetate to fix dyes to fibers made it invaluable for producing richly colored textiles, especially in the wool industry. Lead acetate is also used in the tanning of leather, where it helps to preserve and soften the leather, enhancing its quality and durability. Despite the fact that many industries have moved away from using lead-based compounds due to safety concerns, lead acetate still plays a role in certain niche markets, particularly in the production of high-quality leather goods. In addition to its use in textiles and leather, lead acetate anhydrous is used in various chemical syntheses. It serves as a reagent in organic chemistry, where it is used in the production of other lead compounds. It is also employed in laboratories for specific reactions, such as in the preparation of lead oxide and in certain types of chemical analyses. Lead acetate's role as a catalyst in some reactions has also been explored, though its use in this capacity is less common today due to the known hazards associated with lead. Lead acetate has been used historically in medicine as well, especially in the treatment of ailments such as sore throats and skin conditions. However, its toxic nature, particularly the dangers of lead poisoning, has led to its discontinuation in medicinal applications. Lead poisoning can occur when lead acetate is ingested or absorbed through the skin, leading to a range of health problems, including neurological damage and kidney failure. Because of these risks, the use of lead acetate in consumer products and medicine has been severely restricted in many countries. One of the most significant applications of lead acetate anhydrous in modern times is its role in laboratory experiments and as an industrial reagent. It is used in the preparation of various lead-based compounds, including lead salts and other intermediates required for the manufacture of lead-containing products. Additionally, lead acetate is still used in some chemical testing processes, such as in the identification of sulfide ions, where it reacts with sulfur compounds to form lead sulfide, a black precipitate. This reaction is used in qualitative analysis in the laboratory. Due to its toxicity, lead acetate is subject to strict regulation in many countries, with guidelines governing its safe use, handling, and disposal. In the European Union and the United States, lead acetate has been classified as a hazardous substance, and its use in consumer products, particularly those that may lead to ingestion or exposure, is prohibited. Alternative, non-toxic chemicals have largely replaced lead acetate in many of its traditional applications, especially in industries where worker safety and environmental concerns are a priority. In conclusion, lead acetate anhydrous is a historically significant compound with a variety of industrial applications, especially in textiles, leather tanning, and chemical synthesis. However, its toxic nature has significantly limited its use in recent decades. While its role in modern industries has diminished due to health concerns, it remains an important compound in specific niches. The discovery of its toxicity has led to increased awareness and regulation of its use, as well as the development of safer alternatives. References 2024. Demyelination and impaired oligodendrogenesis in the corpus callosum following lead exposure. Toxicological sciences : an official journal of the Society of Toxicology, 201. DOI: 10.1093/toxsci/kfae100 2012. Enhanced nitric oxide production during lead (Pb2+) exposure recovers protein expression but not presynaptic localization of synaptic proteins in developing hippocampal neurons. Brain Research, 1440. DOI: 10.1016/j.brainres.2011.12.037 1984. Electrocorticogram (ECoG) and auditory evoked potential (AEP) in rats intoxicated with lead acetate. INDUSTRIAL HEALTH, 22. DOI: 10.2486/indhealth.22.189 |
| Market Analysis Reports |