Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic copper |
|---|---|
| Name | Copper(II) hexafluoroacetylacetonate hydrate |
| Synonyms | (SP-4-1)-Bis(1,1,1,5,5,5-hexafluoro-2,4-pentanedionato-O,O')copper hydrate |
| Molecular Structure | 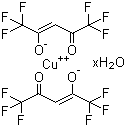 |
| Molecular Formula | C10H2CuF12O4.xH2O |
| CAS Registry Number | 155640-85-0 |
| EC Number | 628-023-7 |
| SMILES | C(=C(C(F)(F)F)[O-])C(=O)C(F)(F)F.C(=C(C(F)(F)F)[O-])C(=O)C(F)(F)F.O.[Cu+2] |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Copper(II) hexafluoroacetylacetonate hydrate is a significant compound in the field of coordination chemistry, valued for its unique properties and wide range of applications. This substance is a coordination complex of copper(II) with hexafluoroacetylacetonate (hfac) as the ligand and water molecules incorporated into the crystal structure. The discovery of Copper(II) hexafluoroacetylacetonate hydrate emerged from the broader study of metal-organic complexes where the aim was to synthesize compounds with specific electronic and structural characteristics. The compound is formed by reacting copper(II) salts with hexafluoroacetylacetone (hfacH), resulting in a complex where the copper center is coordinated by the hfac ligands and additional water molecules. The hydrate form involves the inclusion of water in the crystal lattice, which can influence the physical properties of the compound. The structure of Copper(II) hexafluoroacetylacetonate hydrate is characterized by a copper(II) ion coordinated in a square planar arrangement with four fluorinated acetylacetonate ligands. The hexafluoroacetylacetonate ligand, known for its strong electron-withdrawing properties due to the fluorine atoms, stabilizes the copper center and imparts distinct electronic characteristics to the complex. The hydrate form contains water molecules that participate in hydrogen bonding and contribute to the stability and solubility of the compound in various solvents. One of the primary applications of Copper(II) hexafluoroacetylacetonate hydrate is in the field of catalysis. The compound is utilized as a catalyst or catalyst precursor in various chemical reactions. Its stability and electronic properties make it suitable for use in oxidation reactions, polymerization processes, and other catalytic transformations. The presence of fluorinated ligands can also enhance the reactivity and selectivity of the catalyst, making it a valuable tool in synthetic chemistry. In addition to its catalytic applications, Copper(II) hexafluoroacetylacetonate hydrate is used in materials science. It serves as a precursor for the synthesis of copper-containing thin films and coatings, which are important in electronics and optical applications. The compound's ability to form stable complexes with other materials allows for the fabrication of high-quality films with controlled properties. Furthermore, the compound is studied for its potential applications in the field of magnetic materials. The strong ligand field created by the hexafluoroacetylacetonate ligand can influence the magnetic properties of the copper center, making it a subject of interest for developing new magnetic materials with tailored characteristics. In summary, Copper(II) hexafluoroacetylacetonate hydrate represents an important class of copper coordination complexes with diverse applications in catalysis, materials science, and magnetic materials. Its unique structural features and properties underscore its significance in both research and industrial applications. References none |
| Market Analysis Reports |