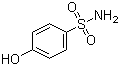
4-Hydroxybenzenesulfonamide, commonly known as sulfanilamide, is an important organic compound with significant historical and practical applications. Characterized by its sulfonamide group attached to a hydroxybenzoic acid moiety, this substance has played a crucial role in the development of antibiotics and continues to be relevant in various fields today.
The discovery of 4-hydroxybenzenesulfonamide dates back to the early 20th century. In 1932, German chemists Gerhard Domagk and colleagues discovered that sulfanilamide, a closely related compound, exhibited remarkable antibacterial properties. This discovery marked the advent of sulfa drugs, the first class of antibiotics used to treat bacterial infections. Sulfanilamide was found to inhibit bacterial growth by interfering with the synthesis of folic acid, a vital nutrient for bacteria. This mechanism of action was revolutionary, providing a new method to combat infections that were previously difficult to treat.
The development of sulfanilamide and its derivatives ushered in a new era of medicine, dramatically improving the treatment of bacterial infections and reducing mortality rates from diseases such as pneumonia, meningitis, and sepsis. The introduction of sulfa drugs in the 1930s and 1940s played a pivotal role in advancing modern medicine and public health.
In addition to its historical significance, 4-hydroxybenzenesulfonamide has applications beyond its use as an antibiotic. The compound serves as a precursor in the synthesis of various other sulfonamide drugs. These derivatives have been used to treat a range of conditions, including bacterial infections, autoimmune diseases, and certain types of cancer. For example, sulfonamide derivatives are used in the treatment of ulcerative colitis and rheumatoid arthritis, showcasing the compound's versatility in medicine.
Moreover, 4-hydroxybenzenesulfonamide has been employed in biochemical research. Its ability to form stable complexes with metal ions makes it useful in studies involving enzyme inhibition and metal-binding properties. Researchers use this compound to explore mechanisms of action and develop new therapeutic agents.
The compound's role extends to materials science as well. Its sulfonamide group can be incorporated into various polymers and resins, contributing to the development of advanced materials with tailored properties. This application highlights the compound's utility beyond the realm of pharmaceuticals, demonstrating its versatility in different scientific and industrial fields.
In summary, 4-hydroxybenzenesulfonamide is a compound of significant historical and practical importance. From its groundbreaking role in the development of sulfa drugs to its diverse applications in research and materials science, this chemical substance continues to impact various fields. Its discovery marked a turning point in medical science, and its ongoing use underscores its continued relevance in modern chemistry and industry.
References
2014. Sulfonamide inhibition studies of two β-carbonic anhydrases from the bacterial pathogen Legionella pneumophila. Bioorganic & Medicinal Chemistry. URL: https://pubmed.ncbi.nlm.nih.gov/24792813
|























 GHS07 Warning Details
GHS07 Warning Details