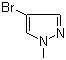
4-Bromo-1-methylpyrazole is a halogenated heterocyclic compound belonging to the pyrazole family. Its molecular structure consists of a five-membered ring containing two adjacent nitrogen atoms (positions 1 and 2), with a bromine atom substituted at the 4-position and a methyl group attached to the nitrogen at the 1-position. The molecular formula of the compound is C4H5BrN2.
The compound is typically prepared through selective halogenation of 1-methylpyrazole, using reagents such as N-bromosuccinimide (NBS) or molecular bromine under controlled conditions. The reaction tends to favor substitution at the 4-position due to the electronic distribution of the pyrazole ring and the directing effect of the N-methyl group. The synthesis may involve intermediate protection or regioselective methods to enhance yield and minimize side reactions.
4-Bromo-1-methylpyrazole serves as an important intermediate in synthetic organic chemistry, especially in the design of pharmacologically active compounds and agrochemical agents. The pyrazole ring is a widely recognized structural motif in medicinal chemistry due to its ability to interact with biological targets such as enzymes, receptors, and nucleic acids. The introduction of a bromine atom enhances the molecule's utility by providing a site for further functionalization via palladium-catalyzed cross-coupling reactions, such as Suzuki–Miyaura or Buchwald–Hartwig couplings.
The presence of the methyl group on the nitrogen atom at position 1 improves the compound’s solubility and alters its electronic properties, affecting reactivity and binding characteristics. The compound is often used to construct more complex molecules by substituting the bromine with aryl, heteroaryl, or alkyl groups, leading to a variety of pyrazole derivatives with potential applications in pharmaceuticals, materials science, and crop protection.
In drug discovery, derivatives of pyrazole—including halogenated and N-substituted variants—have been evaluated for activities such as anti-inflammatory, anticancer, antifungal, and kinase inhibitory effects. Although 4-bromo-1-methylpyrazole itself is not widely reported as a final drug molecule, it functions as a versatile scaffold in the exploration of structure–activity relationships during lead optimization.
In materials chemistry, pyrazole-based ligands are often used to coordinate metal ions, and halogenated pyrazoles like 4-bromo-1-methylpyrazole can be modified to create polydentate ligands for catalysis or coordination polymers. The electron-withdrawing effect of the bromine atom influences the acidity of nearby hydrogen atoms and the electron density distribution across the ring, which can be advantageous in ligand design.
The compound is a colorless to pale yellow liquid or crystalline solid, depending on purity and storage conditions. It is soluble in common organic solvents such as ethanol, acetone, and dichloromethane. Its structure can be confirmed by spectroscopic techniques: 1H and 13C nuclear magnetic resonance (NMR) spectroscopy show characteristic shifts for the ring protons and methyl group; infrared (IR) spectroscopy displays N–H or C–H stretches and ring vibrations; and mass spectrometry confirms the molecular ion and isotopic pattern associated with bromine.
Safety data indicate that 4-bromo-1-methylpyrazole should be handled with standard laboratory precautions. Halogenated heterocycles may cause irritation to the eyes, skin, or respiratory tract. Avoidance of direct contact and inhalation, along with use of personal protective equipment, is advised. The compound should be stored in a tightly sealed container in a cool, dry place away from light and moisture.
In conclusion, 4-bromo-1-methylpyrazole is a useful synthetic intermediate that combines the reactive halogen functionality with the pyrazole core structure. Its applications span pharmaceutical and agrochemical synthesis, as well as coordination and materials chemistry, making it a valuable building block in research and development.
References
2020. The Problem with Problems: Fundamental to Applied Research Using Palladium. Synlett, 31(9).
DOI: 10.1055/s-0039-1690904
2018. Convenient Synthesis of 5-Aryl-1-(1H-pyrazol-4-yl)pyrrolidin-2-ones. Russian Journal of Organic Chemistry, 54(6).
DOI: 10.1134/s1070428018060234
2017. Pyrazole-4-boronic Acids from 4-Bromopyrazoles. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-112-00305
|































































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details