Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Aryl compounds >> Naphthalenes |
|---|---|
| Name | 2-Acetyl-6-bromonaphthalene |
| Synonyms | 1-(6-bromonaphthalen-2-yl)ethanone |
| Molecular Structure | 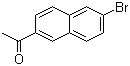 |
| Molecular Formula | C12H9BrO |
| Molecular Weight | 247.98 |
| CAS Registry Number | 1590-25-6 |
| SMILES | CC(=O)C1=CC2=C(C=C1)C=C(C=C2)Br |
| Density | 1.5±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 357.1±15.0 °C 760 mmHg (Calc.)* |
| Flash point | 104.2±7.7 °C (Calc.)* |
| Index of refraction | 1.641 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
2-Acetyl-6-bromonaphthalene is an organic compound that belongs to the class of halogenated naphthalene derivatives. It has the chemical formula C10H9BrO, and its structure consists of a naphthalene ring with two functional groups: an acetyl group (-COCH3) at position 2 and a bromine atom at position 6 of the naphthalene ring. The synthesis of 2-acetyl-6-bromonaphthalene typically involves halogenation and subsequent acylation reactions. In the laboratory, it can be synthesized through the bromination of naphthalene followed by acylation with acetic acid or an acylating agent such as acetyl chloride. This compound has been studied for its role in various chemical and industrial applications. It is commonly used as an intermediate in the synthesis of more complex organic molecules, including potential pharmaceuticals, agrochemicals, and other specialty chemicals. Due to its functional groups, it can undergo a variety of chemical reactions, such as nucleophilic substitution, Friedel-Crafts acylation, and reactions involving the bromine atom for further functionalization or derivatization. While its direct applications in pharmaceuticals or other fields may not be as prominent as some other naphthalene derivatives, compounds like 2-acetyl-6-bromonaphthalene are valuable in the development of specific chemical libraries for research and as building blocks for the synthesis of more complex organic materials. Additionally, the presence of both an acetyl group and a bromine atom provides versatility for further modifications to enhance biological or chemical properties. In the context of research, halogenated naphthalene derivatives, including 2-acetyl-6-bromonaphthalene, can be used as starting materials for the design of organic semiconductors, liquid crystals, or materials with optoelectronic properties. The bromine atom in particular can be useful for introducing crosslinking capabilities or other functionalizations to modify the chemical reactivity of the compound. In summary, 2-acetyl-6-bromonaphthalene is a versatile organic intermediate used in synthetic chemistry. Its functional groups allow for further chemical modifications, making it valuable in the development of more complex compounds for industrial, pharmaceutical, and research applications. References none |
| Market Analysis Reports |