Online Database of Chemicals from Around the World
| Shanghai Sinofluoro chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinofluoro.com | |||
 | +86 (21) 6427-9360 | |||
 | +86 (21) 6427-8603 | |||
 | simon.lv@sinofluoro.com jerry.chen@sinofluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sanming Hexafluo Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hexafluo.com | |||
 | +86 (598) 281-7768 | |||
 | +86 (598) 286-6778 | |||
 | info@hexafluo.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Chuan Shuo Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.transol.cn | |||
 | +86 (571) 8760-9021 | |||
 | qian@transol.cn | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Organic fluorine compound |
|---|---|
| Name | Perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride |
| Synonyms | 1,1,2,2-Tetrafluoro-2-[1,2,2-trifluoro-1-(trifluoromethyl)-2-[(trifluorovinyl)oxy]ethoxy]ethanesulfonyl fluoride |
| Molecular Structure | 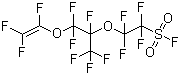 |
| Molecular Formula | C7F14O4S |
| Molecular Weight | 446.11 |
| CAS Registry Number | 16090-14-5 |
| EC Number | 240-249-4 |
| SMILES | C(=C(F)F)(OC(C(C(F)(F)F)(OC(C(F)(F)S(=O)(=O)F)(F)F)F)(F)F)F |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H336 Details | ||||||||||||
| Safety Statements | P261-P271-P272-P280-P302+P352-P304+P340-P319-P321-P333+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride is a specialized fluorinated organic compound that features a sulfonyl fluoride functional group attached to a perfluorinated ether-containing alkene backbone. It belongs to the class of perfluoroalkylsulfonyl fluorides, which are known for their high thermal stability, chemical inertness, and utility in materials chemistry, particularly in the development of ion-exchange membranes and specialty polymers. The molecular structure consists of a terminal sulfonyl fluoride group (–SO2F) connected to a chain containing fully fluorinated carbon atoms and ether linkages (–CF2O–), with a vinyl-like terminal perfluoroalkene moiety. The presence of two ether groups and a branching methyl group in the backbone increases flexibility and modifies the electronic properties of the molecule. The perfluorinated chain significantly reduces the compound’s surface energy and enhances its resistance to chemical degradation. Perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride is closely related to compounds used as monomers or intermediates in the synthesis of perfluorosulfonic acid ionomers such as Nafion®. These polymers are widely used in applications like proton exchange membrane fuel cells (PEMFCs), chlor-alkali electrolysis, and chemical-resistant coatings. The sulfonyl fluoride group is a precursor to the sulfonic acid functionality found in ionomer membranes, and it can be hydrolyzed or converted into a variety of sulfonated derivatives depending on the process conditions. The synthesis of this compound typically involves telomerization or fluorination reactions starting from simpler perfluoroalkyl precursors. Perfluoroalkylvinyl ethers are key intermediates, and the introduction of the sulfonyl fluoride group is generally achieved by electrophilic sulfonation followed by fluorination. These reactions require specialized fluorination reagents and must be conducted under strictly controlled, anhydrous conditions due to the moisture sensitivity of sulfonyl fluorides. One of the distinguishing features of perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride is its capacity to undergo free radical polymerization via the terminal alkene group. This reactivity enables the molecule to be co-polymerized with tetrafluoroethylene (TFE) or other fluorinated monomers to produce high-performance fluoropolymers. Such polymers combine mechanical strength and flexibility with exceptional resistance to acids, bases, and solvents, making them suitable for use in chemically aggressive environments. Analytical identification and characterization of the compound rely on techniques such as nuclear magnetic resonance (NMR) spectroscopy, particularly 19F NMR, to observe the distinct chemical shifts of fluorine atoms in various environments. Infrared (IR) spectroscopy is used to confirm the presence of the sulfonyl fluoride group, with characteristic S=O and S–F stretching bands. Mass spectrometry and elemental analysis may also be employed for molecular confirmation and purity assessment. Due to the presence of the sulfonyl fluoride group, the compound is electrophilic and reactive toward nucleophilic substitution. It can react with amines, alcohols, or water under appropriate conditions to form sulfonamides, sulfonate esters, or sulfonic acids, respectively. These transformations are essential steps in modifying or activating the compound for subsequent processing in membrane fabrication or surface treatment applications. Regarding handling and safety, perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride should be managed using standard laboratory precautions. The sulfonyl fluoride group may cause skin and eye irritation and should not be inhaled or ingested. Perfluorinated compounds are also of environmental concern due to their persistence and potential for bioaccumulation. Waste materials and residues should be disposed of in compliance with environmental regulations. In summary, perfluoro(4-methyl-3,6-dioxaoct-7-ene)sulfonyl fluoride is an important intermediate used in the synthesis of fluorinated ionomers and specialty polymers. Its combination of perfluorinated ether groups and a reactive sulfonyl fluoride functionality enables it to serve as a valuable building block in the manufacture of advanced materials for fuel cells, membranes, and chemically resistant coatings. References 2014. Radical Copolymerization of Fluorinated Functional Monomers with 1,1-Difluoroethene. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-124-00144 2006. [2+2]Cycloadditions. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-024-00185 2021. Updatable List to add PFAS Structures to Public Resources from ECI (UniLu). Zenodo. DOI: 10.5281/zenodo.6389740 |
| Market Analysis Reports |