Online Database of Chemicals from Around the World
| Tikslioji Sinteze | Lithuania | |||
|---|---|---|---|---|
 | www.finesynthesis.lt | |||
 | +370 (5) 213-5553 | |||
 | ardan@micro.lt | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Chongqing Xingcan Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.starrymed.com | |||
 | +86 (23) 6120-3303 +86 13650506873 | |||
 | +86 (23) 6120-3303 | |||
 | xingcanyaoye@sina.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Chemical reagent >> Organic reagent >> Alkane |
|---|---|
| Name | 4,4'-Di-tert-butylbiphenyl |
| Synonyms | 4,4'-Di-tert-butyl-1,1'-biphenyl |
| Molecular Structure | 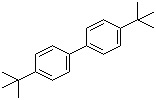 |
| Molecular Formula | C20H26 |
| Molecular Weight | 266.42 |
| CAS Registry Number | 1625-91-8 |
| EC Number | 216-615-4 |
| SMILES | CC(C)(C)C1=CC=C(C=C1)C2=CC=C(C=C2)C(C)(C)C |
| Melting point | 126-129 °C |
|---|---|
| Boiling point | 190-192 °C (13 mmHg) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4,4'-Di-tert-butylbiphenyl is an organic compound that has gained importance due to its stability and versatility in various chemical and industrial applications. The discovery of 4,4'-di-tert-butylbiphenyl can be linked to the exploration of substituted biphenyls, a class of compounds characterized by two connected benzene rings. The introduction of bulky tert-butyl groups at the 4 and 4' positions of the biphenyl structure enhances the compound’s stability by hindering oxidative and thermal degradation, making it particularly useful in demanding environments. The synthesis of 4,4'-di-tert-butylbiphenyl typically involves Friedel-Crafts alkylation, a process where biphenyl is reacted with tert-butyl chloride in the presence of a Lewis acid catalyst, such as aluminum chloride. This reaction efficiently introduces the tert-butyl groups onto the biphenyl core, resulting in a compound with high purity and stability. The steric effects of the tert-butyl groups prevent the rings from easily undergoing further substitution or unwanted reactions, preserving the integrity of the molecule under various conditions. One of the primary applications of 4,4'-di-tert-butylbiphenyl is as a stabilizer in the production of polymers and plastics. The compound acts as an antioxidant, preventing the degradation of polymers during processing and prolonging the lifespan of the final products. Its effectiveness in stabilizing polymers makes it a key additive in the manufacturing of high-performance materials, such as those used in the automotive and aerospace industries, where durability and resistance to environmental factors are crucial. In addition to its role as a stabilizer, 4,4'-di-tert-butylbiphenyl is used in the formulation of lubricants and greases. The compound’s stability at high temperatures and its ability to resist oxidation make it an ideal component in lubricants designed for use in extreme conditions. By preventing the breakdown of the lubricant, 4,4'-di-tert-butylbiphenyl helps maintain the performance of machinery and engines, reducing wear and extending the service life of critical components. The electronic industry also benefits from the use of 4,4'-di-tert-butylbiphenyl, particularly in the development of organic semiconductors and insulating materials. The compound’s rigid biphenyl core and bulky tert-butyl groups contribute to its effectiveness as an insulating layer in organic electronic devices. These properties are valuable in applications such as organic light-emitting diodes (OLEDs) and organic field-effect transistors (OFETs), where precise control over the electronic properties of materials is essential for device performance. Research into 4,4'-di-tert-butylbiphenyl continues to explore its potential in new applications, including its use as a precursor for more complex organic molecules. The compound’s stable structure serves as a foundation for the synthesis of derivatives that could have enhanced properties for specific applications. For instance, modifying the biphenyl core or the tert-butyl groups could lead to the development of new materials with tailored properties for use in advanced technologies. Furthermore, 4,4'-di-tert-butylbiphenyl is being investigated for its potential use in environmental applications. Its stability and low reactivity make it a candidate for use in processes that require inert conditions, such as the storage or transportation of reactive chemicals. Additionally, the compound’s hydrophobic nature allows it to be used in the formulation of water-resistant coatings and sealants, contributing to the protection of materials exposed to moisture and harsh environments. As industries continue to seek materials that offer both stability and versatility, 4,4'-di-tert-butylbiphenyl is likely to remain an important compound in various fields. Its combination of thermal stability, oxidative resistance, and chemical inertness ensures its continued relevance in applications ranging from polymer stabilization to electronic device manufacturing. Ongoing research and development are expected to further expand the use of 4,4'-di-tert-butylbiphenyl in emerging technologies and industries. References 2021. Synthesis of five-membered nitrogen-containing heterocycles using copper. Journal of the Iranian Chemical Society, 18(12), 3205-3224. DOI: 10.1007/s13738-021-02350-5 2017. Electrodeposition of 4,4′-di-tert-butylbiphenyl peroxide from the anodic oxidation of p-tert-butylphenol in an alkaline acetonitrile solution. Journal of Applied Electrochemistry, 47(4), 453-463. DOI: 10.1007/s10800-016-1041-2 2012. Synthesis and some transformations of 2- and 2,2′-substituted bis(ethylenedioxy)biphenyls containing cyclopropane fragments. Russian Journal of Organic Chemistry, 48(1), 61-70. DOI: 10.1134/s107042801201006x |
| Market Analysis Reports |