Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Huangshi Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huangshipharma.com | |||
 | +86 (714) 329-3589 | |||
 | xiebihuan@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Nanjing Pharma Chemical Plant | China | |||
|---|---|---|---|---|
 | www.njpharmchem.com | |||
 | +86 (25) 8557-3482 | |||
 | +86 (25) 8557-9530 | |||
 | njpc@163.com | |||
| Chemical manufacturer since 1995 | ||||
| OChem Incorporation | USA | |||
|---|---|---|---|---|
 | www.ocheminc.com | |||
 | +1 (847) 403-7044 | |||
 | +1 (847) 298-5008 | |||
 | sales@ocheminc.com | |||
| Chemical manufacturer | ||||
| SynInnova Laboratories Inc. | USA | |||
|---|---|---|---|---|
 | www.syninnova.com | |||
 | +1 (780) 485-2962 | |||
 | +1 (780) 485-2963 | |||
 | sales@syninnova.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Furan and pyran |
|---|---|
| Name | Furazidine |
| Synonyms | 1-((3-(5-Nitro-2-furyl)allylidene)amino)hydantoin; 1-[[(E)-3-(5-Nitrofuran-2-yl)prop-2-enylidene]amino]imidazolidine-2,4-dione |
| Molecular Structure | 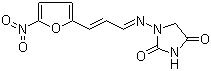 |
| Molecular Formula | C10H8N4O5 |
| Molecular Weight | 264.19 |
| CAS Registry Number | 1672-88-4 |
| EC Number | 681-332-9 |
| SMILES | C1C(=O)NC(=O)N1/N=C/C=C/C2=CC=C(O2)[N+](=O)[O-] |
| Density | 1.6±0.1 g/cm3 Calc.* |
|---|---|
| Solubility | DMSO: 47mg/mL (Expl.) |
| Index of refraction | 1.689 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Furazidine is a synthetic nitrofuran derivative known chemically as 1-[(5-nitrofuran-2-yl)methylideneamino]imidazolidin-2-one. It belongs to the class of nitrofuran antimicrobials, compounds characterized by a nitrofuran ring system attached to various functional groups that contribute to their antibacterial properties. Furazidine’s molecular structure includes a nitrofuran moiety linked via an imine (Schiff base) bond to an imidazolidinone ring, imparting distinct chemical and pharmacological characteristics. The compound was developed as part of efforts to optimize nitrofuran drugs, which have been used since the mid-20th century for their broad-spectrum activity against Gram-positive and Gram-negative bacteria. Furazidine was designed to improve antimicrobial efficacy and reduce toxicity relative to earlier nitrofurans. Its unique structure contributes to its pharmacokinetic and pharmacodynamic properties. Furazidine is primarily used as an antibacterial agent, especially in the treatment of urinary tract infections (UTIs). The drug’s activity results from its bioreduction within bacterial cells, where the nitro group undergoes enzymatic reduction, generating reactive intermediates that damage bacterial DNA, proteins, and other macromolecules, leading to cell death. This mechanism is typical of nitrofuran derivatives and contributes to their bactericidal effects. The compound has been employed clinically in several countries for the management of uncomplicated urinary tract infections, where it offers an oral treatment option with relatively low rates of resistance compared to other antibiotics. Its spectrum includes common uropathogens such as Escherichia coli and Staphylococcus saprophyticus. Due to its mode of action, furazidine exhibits activity against both aerobic and anaerobic bacteria. Furazidine’s pharmacological profile includes good oral bioavailability and renal excretion, allowing it to achieve effective concentrations in the urinary tract. It is generally well tolerated, although adverse effects such as gastrointestinal discomfort and hypersensitivity reactions can occur. Its use requires careful adherence to dosage and duration to minimize the risk of resistance development and side effects. In synthesis, furazidine can be prepared by condensation of 5-nitrofuraldehyde with imidazolidin-2-one, forming the Schiff base linkage. This straightforward synthetic route enables the production of furazidine with high purity and yield, facilitating its pharmaceutical formulation. Analytical characterization of furazidine involves several techniques. Proton nuclear magnetic resonance (²H NMR) spectroscopy confirms the structure, showing characteristic aromatic protons from the nitrofuran ring and the imidazolidinone moiety. Infrared (IR) spectroscopy identifies functional groups such as nitro (NO2) stretches near 1500 and 1350 cm−1, imine (C=N) absorption bands, and carbonyl (C=O) stretches of the imidazolidinone. Mass spectrometry provides molecular weight confirmation and fragmentation pattern consistent with the compound’s structure. Physically, furazidine is typically obtained as a crystalline solid with limited solubility in water but good solubility in organic solvents like dimethyl sulfoxide (DMSO) and ethanol. Its stability and formulation characteristics make it suitable for oral dosage forms such as tablets. In summary, furazidine is a nitrofuran-based antimicrobial agent with a specific application in treating urinary tract infections. Its chemical structure features a nitrofuran ring linked to an imidazolidinone via a Schiff base, contributing to its antibacterial activity through bioreductive damage to bacterial components. Furazidine’s synthesis, pharmacology, and clinical use exemplify its role as a valuable drug in antimicrobial therapy. References 2020. The antibiotic furagin and its derivatives are isoform-selective human carbonic anhydrase inhibitors. Journal of Enzyme Inhibition and Medicinal Chemistry, 35(1). DOI: 10.1080/14756366.2020.1752201 2016. Multi-technique approach for qualitative and quantitative characterization of furazidin degradation kinetics under alkaline conditions. Journal of Pharmaceutical and Biomedical Analysis, 129. DOI: 10.1016/j.jpba.2016.07.039 2008. Ciprofloxacin and furagin in acute cystitis: comparison of early immune and microbiological results. International Journal of Antimicrobial Agents, 31(2). DOI: 10.1016/j.ijantimicag.2007.08.021 |
| Market Analysis Reports |