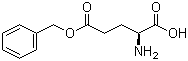
gamma-Benzyl L-glutamate is a derivative of the naturally occurring amino acid L-glutamic acid, where the gamma-carboxyl group is protected by a benzyl ester. This chemical modification is commonly used in peptide synthesis and organic chemistry to selectively protect the side chain of glutamic acid, allowing for controlled reactions on other functional groups without interference.
The compound was developed as part of advancements in synthetic peptide chemistry, which sought effective methods to protect reactive groups during multi-step synthesis. The benzyl ester protecting group was chosen due to its stability under a variety of reaction conditions and its ability to be selectively removed through catalytic hydrogenation, preserving the integrity of the peptide chain or other molecular structures.
In practice, gamma-Benzyl L-glutamate is widely used as an intermediate in the synthesis of peptides and peptidomimetics, particularly where the glutamic acid residue must be selectively protected to prevent side reactions. This selectivity facilitates the formation of peptide bonds at the amino and alpha-carboxyl groups while the gamma-carboxyl remains temporarily masked. After the desired peptide assembly, the benzyl protecting group can be removed under mild hydrogenation conditions, regenerating the free gamma-carboxyl functionality.
Beyond peptide synthesis, gamma-Benzyl L-glutamate is also utilized in the preparation of glutamic acid derivatives for pharmaceutical research. These derivatives serve as building blocks in drug discovery programs targeting enzymes or receptors that interact with glutamate or its analogs. The ability to introduce specific modifications at the gamma position while maintaining the overall stereochemistry of the amino acid is crucial for designing compounds with enhanced biological activity or improved pharmacokinetic properties.
The compound has also found use in material science and chemical biology, where glutamate derivatives are employed in the design of novel biomaterials and molecular probes. The benzyl protection strategy allows for the incorporation of glutamate residues into complex molecular architectures without undesired side reactions, enabling the creation of functionalized polymers, hydrogels, and other bioconjugates.
Synthetically, gamma-Benzyl L-glutamate can be prepared by esterification of L-glutamic acid with benzyl alcohol under acidic conditions. Alternative methods include using benzyl chloride or benzyl bromide with appropriate bases to achieve the selective protection at the gamma-carboxyl group. The stereochemical purity is generally maintained throughout the synthesis, which is essential for applications involving biological systems where chirality plays a key role.
The protective benzyl group is stable against bases and nucleophiles but can be efficiently removed by catalytic hydrogenation using palladium catalysts under mild conditions. This feature makes gamma-Benzyl L-glutamate a versatile intermediate that fits well into solid-phase peptide synthesis protocols and other synthetic strategies requiring temporary protection.
Overall, gamma-Benzyl L-glutamate represents a fundamental tool in modern synthetic chemistry, especially in the preparation of peptides and biologically active molecules. Its discovery and widespread use have greatly facilitated the synthesis of complex molecules, enhancing the ability to explore protein function, develop new therapeutics, and create innovative biomaterials. Its combination of chemical stability and ease of deprotection underscores its enduring importance in chemical and pharmaceutical research.
References
2014. Direct Synthesis of Polybenzylated Glutamic Acid Monoesters: Disambiguation of N,N-Dibenzylglutamic Acid α- and γ-Benzyl Esters. Synlett, 25(16).
DOI: 10.1055/s-0034-1378532
2015. Synthesis and structure formation of block copolymers of poly(ethylene glycol) with homopolymers and copolymers of l-glutamic acid Γ-benzyl ester and l-leucine in water. Colloid and Polymer Science, 293(9).
DOI: 10.1007/s00396-015-3632-6
2021. pH-dependent delivery of chlorhexidine from PGA grafted mesoporous silica nanoparticles at resin-dentin interface. Journal of Nanobiotechnology, 19(1).
DOI: 10.1186/s12951-021-00788-6
|









































































 GHS07 Warning Details
GHS07 Warning Details