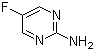
5-Fluoro-2-pyrimidinamine is a chemical compound that belongs to the class of pyrimidine derivatives, specifically a halogenated pyrimidine. This compound features a fluorine atom at the 5-position and an amine group at the 2-position of the pyrimidine ring. The pyrimidine structure itself is a six-membered ring consisting of alternating nitrogen and carbon atoms, a common motif in many biologically active molecules.
The discovery of 5-fluoro-2-pyrimidinamine and similar halogenated pyrimidine derivatives is rooted in the broad interest in modifying nucleobase-like structures for their potential biological activity. Halogenated pyrimidines have been studied for their ability to interact with biological systems, particularly as potential intermediates in the development of pharmaceutical agents. Fluorine substitution is known to influence the electronic properties of organic compounds, often enhancing their stability and bioavailability. The addition of a fluorine atom can also improve the molecule's binding affinity to specific targets, which is a significant advantage in drug development.
This compound and its derivatives have been explored in various research contexts, primarily due to their relevance in medicinal chemistry and the design of therapeutic agents. The incorporation of fluorine into pyrimidine derivatives has been associated with enhanced pharmacological properties, such as improved absorption, distribution, and metabolic stability. In particular, fluoropyrimidines are important in the development of antimetabolite drugs, which interfere with cellular processes by mimicking naturally occurring molecules involved in nucleic acid metabolism.
5-Fluoro-2-pyrimidinamine, due to its structure, may be studied for its potential activity in inhibiting enzymes involved in nucleic acid synthesis or for its role as a building block in the synthesis of more complex medicinal compounds. Pyrimidine derivatives are also valuable in the development of anticancer drugs, antiviral agents, and agents used in the treatment of bacterial infections.
This compound's exact applications would depend on its interaction with specific biological targets, which can be explored through further experimental research. The ability to modify the pyrimidine ring with different substituents, such as halogens, has led to the development of a variety of compounds that can be fine-tuned for specific therapeutic purposes.
In summary, 5-fluoro-2-pyrimidinamine is an important intermediate in the design of halogenated pyrimidine derivatives with potential applications in medicinal chemistry. The fluorine substitution at the 5-position offers unique advantages in terms of stability and bioactivity, making it a promising compound for further exploration in drug development.
References
2021. Electrophilic and Oxidative Fluorination of Heterocyclic Compounds: Contribution to Green Chemistry. Russian Journal of Organic Chemistry, 57(9).
DOI: 10.1134/s1070428021090013
|
















 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details