Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Zhejiang Kangfeng Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kangfeng.com | |||
 | +86 (579) 8670-9298 8670-9155 | |||
 | +86 (579) 8670-9678 | |||
 | wukang@kangfeng.com qimaoxin@kangfeng.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changsha Huajing Powdery Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.her8.cn | |||
 | +86 (731) 8887-9686 | |||
 | +86 (731) 8883-6388 | |||
 | webmaster@her8.cn | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal fluorides and salts |
|---|---|
| Name | Sodium hexafluoroantimonate |
| Molecular Structure | 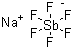 |
| Molecular Formula | NaSbF6 |
| Molecular Weight | 258.74 |
| CAS Registry Number | 16925-25-0 |
| EC Number | 240-989-8 |
| SMILES | F[Sb-](F)(F)(F)(F)F.[Na+] |
| Density | 3.38 g/mL |
|---|---|
| Water solubility | SOLUBLE |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H332-H411 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P273-P301+P317-P304+P340-P317-P330-P391-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Sodium hexafluoroantimonate is an inorganic salt composed of sodium cations and hexafluoroantimonate anions, with the chemical formula NaSbF6. It belongs to the family of hexafluorometallate salts, in which a central metal atom is octahedrally coordinated by six fluoride ligands. Sodium hexafluoroantimonate typically appears as a white to colorless crystalline solid. It is generally insoluble or only sparingly soluble in water, while showing limited solubility in some polar inorganic solvents under specific conditions. The compound is valued primarily for the chemical stability and weakly coordinating nature of the hexafluoroantimonate anion. The development of sodium hexafluoroantimonate is closely connected to the broader study of antimony fluorides and complex fluorometallate salts during the twentieth century. As inorganic and physical chemists explored strong Lewis acids and weakly coordinating anions, hexafluoroantimonate emerged as an important species due to its high symmetry and low basicity. These properties made salts containing the SbF6⁻ anion particularly useful in stabilizing highly reactive cations and intermediates without participating directly in chemical reactions. Sodium hexafluoroantimonate became a convenient and relatively stable source of the hexafluoroantimonate anion for laboratory and industrial use. Synthesis of sodium hexafluoroantimonate is typically achieved by neutralization reactions involving hexafluoroantimonic acid or by metathesis between a sodium salt and another hexafluoroantimonate compound. One common route involves the reaction of antimony pentafluoride with a fluoride source to generate hexafluoroantimonic acid, followed by neutralization with a sodium base to yield the sodium salt. Alternatively, sodium hexafluoroantimonate can be prepared by precipitation from solution when sodium ions are introduced into a system containing soluble hexafluoroantimonate anions. The product is isolated as a crystalline solid and purified by washing and drying to remove residual acids or by-products. Chemically, sodium hexafluoroantimonate is characterized by the remarkable stability of the SbF6⁻ anion. The strong antimony–fluorine bonds and the symmetrical octahedral geometry result in a weakly coordinating anion that interacts minimally with cations. This property is central to its use in chemistry, as it allows the stabilization of highly electrophilic or reactive cationic species without significant anion interference. The salt is generally resistant to hydrolysis under neutral conditions, though strong bases or prolonged exposure to moisture and heat can lead to slow decomposition. In practical applications, sodium hexafluoroantimonate is used primarily as a source of the hexafluoroantimonate anion in research and industrial chemistry. It is employed in the preparation of ionic compounds, catalysts, and advanced materials where a non-nucleophilic, weakly coordinating counterion is required. Such applications include studies of carbocations, organometallic complexes, and superacid chemistry. Sodium hexafluoroantimonate is also used in electrochemical research and in the development of specialized electrolytes, where the stability and low reactivity of the anion contribute to controlled ionic behavior. From a physical standpoint, sodium hexafluoroantimonate is thermally stable under normal laboratory conditions and does not exhibit significant volatility. It should be handled with care due to the presence of antimony and fluoride, both of which can pose health hazards if mishandled. Appropriate protective equipment, including gloves and eye protection, is recommended, and the compound should be stored in tightly sealed containers to avoid moisture uptake and contamination. Overall, sodium hexafluoroantimonate is an important inorganic salt whose significance lies not in bulk applications but in its specialized chemical role. Its ability to provide a stable, weakly coordinating hexafluoroantimonate anion has made it a valuable tool in inorganic, organometallic, and physical chemistry. The compound exemplifies how carefully designed inorganic salts can enable the study and application of highly reactive chemical species by offering stability without unwanted chemical interference. References 2025. Atomic‐level insights into the synergistic effect between ligands on electrochemical CO2 reduction: based on Au7Ag8 series nanoclusters. Rare Metals. DOI: 10.1007/s12598-025-03267-1 |
| Market Analysis Reports |