Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Lanzhou Kohn & Shawn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kyxpharma.com | |||
 | +86 (931) 419-0454 +86 13893344397 | |||
 | +86 (931) 850-6154 | |||
 | sales@kyxpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hebei Delongtai Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dltchem.com | |||
 | +86 (311) 8550-0818 | |||
 | +86 (311) 8550-0817 | |||
 | zhan@dltchem.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Indoles |
|---|---|
| Name | 5,6-Difluoroindole-2-carboxylic acid |
| Synonyms | 5,6-difluoro-1H-indole-2-carboxylic acid |
| Molecular Structure | 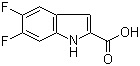 |
| Molecular Formula | C9H5F2NO2 |
| Molecular Weight | 197.14 |
| CAS Registry Number | 169674-35-5 |
| EC Number | 860-786-3 |
| SMILES | C1=C2C=C(NC2=CC(=C1F)F)C(=O)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
5,6-Difluoroindole-2-carboxylic acid is a fluorinated derivative of indole, a fundamental building block of organic chemistry and a key component of a wide range of biologically active compounds. The compound is characterized by the substitution of the indole ring with fluorine atoms at the 5 and 6 positions and a carboxylic acid group at the 2 position. It was first synthesized during the development of novel fluorinated indoles with potential therapeutic applications, using the fluorine atom to impart unique properties. The synthesis of 5,6-difluoroindole-2-carboxylic acid is a multistep reaction starting from appropriately substituted anilines. The common synthetic route involves nitration of the aniline derivative followed by reduction and cyclization to form the indole ring. The introduction of the fluorine atom is achieved by selective fluorination, and the carboxylic acid group is introduced by a carboxylation process. Advanced techniques such as electrophilic fluorination can ensure the precise positioning of the fluorine atom. 5,6-Difluoroindole-2-carboxylic acid shows great potential in medicinal chemistry and other scientific fields due to its unique structure and biological activities associated with fluorinated compounds. This compound is a valuable building block for the synthesis of various drugs. Fluorine atoms are known to enhance the metabolic stability and bioavailability of drugs. 5,6-Difluoroindole-2-carboxylic acid is used to develop inhibitors of enzymes such as kinases and proteases, which are key targets for cancer treatment and other diseases. Its derivatives have shown promise as potential therapeutic agents in preclinical studies. In chemical biology, 5,6-difluoroindole-2-carboxylic acid is a key intermediate for labeling biomolecules with fluorine. This makes it possible to study biological processes using techniques such as fluorine-19 nuclear magnetic resonance (NMR) spectroscopy. The introduction of fluorine atoms helps track molecular interactions and dynamics in complex biological systems. Incorporating fluorine atoms into organic molecules such as indole can significantly change their electronic properties. 5,6-Difluoroindole-2-carboxylic acid is used to design new materials with specific electronic and optical properties that can be used to develop organic semiconductors and light-emitting diodes (LEDs). Ongoing research on 5,6-difluoroindole-2-carboxylic acid is expected to broaden its application. Future studies will focus on optimizing its synthesis, improving its pharmacokinetic properties, and exploring its potential in new therapeutic areas. In addition, the development of novel derivatives and analogs can expand their utility in medicinal chemistry and materials science. References 2022. Novel MreB inhibitors with antibacterial activity against Gram (-) bacteria. Medicinal Chemistry Research, 31(10), 1789-1798. DOI: 10.1007/s00044-022-02967-y |
| Market Analysis Reports |