Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Monils Chemical Engineering Science & Technology (shanghai) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ilschem.cn | |||
 | +86 (21) 3822-8895 | |||
 | +86 (21) 3822-8591 | |||
 | heqing@ilschem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Ionic Liquids Technologies GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.iolitec.de | |||
 | +49 (7666) 913-929 | |||
 | +49 (7666) 912-9345 | |||
 | info@iolitec.de | |||
| Chemical manufacturer | ||||
| Kishida Chemical Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.kishida.co.jp | |||
 | +81 (6) 6946-8061 | |||
 | +81 (6) 6946-1607 | |||
 | shiyaku@kishida.co.jp | |||
| Chemical manufacturer | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| abcr GmbH | Germany | |||
|---|---|---|---|---|
 | www.abcr.de | |||
 | +49 (721) 950-610 | |||
 | +49 (721) 950-6180 | |||
 | info@abcr.de | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Ionic liquid |
|---|---|
| Name | 1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide |
| Synonyms | bis(trifluoromethylsulfonyl)azanide;1-butyl-3-methylimidazol-3-ium |
| Molecular Structure | 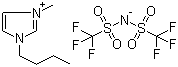 |
| Molecular Formula | C10H15F6N3O4S2 |
| Molecular Weight | 419.36 |
| CAS Registry Number | 174899-83-3 |
| EC Number | 605-742-4 |
| SMILES | CCCCN1C=C[N+](=C1)C.C(F)(F)(F)S(=O)(=O)[N-]S(=O)(=O)C(F)(F)F |
| Refractive index | 1.428 |
|---|---|
| Flash point | >200 °C |
| Hazard Symbols |     GHS05;GHS06;GHS08;GHS09 Danger Details GHS05;GHS06;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301+H311-H301-H311-H314-H373-H411 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P262-P264-P270-P273-P280-P301+P316-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P319-P321-P330-P361+P364-P363-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2922 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide (BMIM TFSI) is a well-known ionic liquid with the molecular formula C9H11F6N3O4S2. It is highly regarded for its excellent properties and is highly valued in a variety of scientific and industrial applications. BMIM TFSI originates from the broader field of ionic liquids, which has received widespread attention since the late 20th century. Researchers sought solvents with enhanced performance characteristics compared to traditional organic solvents, such as low volatility, high thermal stability, and a wide electrochemical window. BMIM TFSI was developed to meet these needs by combining the 1-butyl-3-methylimidazolium cation with the bis(trifluoromethylsulfonyl)imide anion. 1-Butyl-3-methylimidazolium chloride (BMIM Cl) reacts with lithium bis(trifluoromethylsulfonyl)imide (LiTFSI) to produce BMIM TFSI and a byproduct lithium chloride (LiCl) precipitate. The process is usually carried out in a suitable solvent or directly in the ionic liquid phase, followed by purification to separate the ionic liquid. BMIM TFSI is widely used in electrochemical applications due to its high ionic conductivity and wide electrochemical stability window. It can be used as an electrolyte in a variety of energy storage devices, including lithium-ion batteries, supercapacitors, and fuel cells. Its low viscosity and thermal stability improve the efficiency and performance of these devices, making it a key component in advanced energy technologies. As a green solvent, BMIM TFSI has significant advantages over traditional organic solvents. It is non-volatile and environmentally friendly, reducing the risks associated with traditional solvents. BMIM TFSI is used in organic synthesis and extraction processes to improve reaction efficiency and selectivity while minimizing environmental impact. In catalysis, BMIM TFSI can be used as both a solvent and a catalyst. It is able to dissolve a wide range of organic and inorganic compounds, becoming a versatile medium for catalytic reactions. BMIM TFSI is used in homogeneous and heterogeneous catalysis to improve reaction rates, yields, and selectivity. Its use helps achieve more sustainable and efficient chemical synthesis. BMIM TFSI is very effective in separation processes, including liquid-liquid extraction and gas absorption. Its unique solvation properties enable the selective extraction and separation of specific compounds from a mixture. This application is valuable in drug purification, metal extraction, and separation of organic compounds to improve process efficiency and selectivity. In materials science, BMIM TFSI is used to synthesize and modify materials with customized properties. It plays a role in the preparation of nanoparticles, polymers, and other advanced materials. BMIM TFSI can act as a template or stabilizer during the synthesis of nanomaterials, affecting their size, shape, and stability, thereby developing new materials with special properties. References 2024. Biocatalytic transesterification of salmon oil in ionic liquid media to obtain concentrates of omega-3 polyunsaturated fatty acids. European Food Research and Technology. DOI: 10.1007/s00217-024-04484-1 2023. Tutorial for thermal analysis of ionic liquids. Journal of Thermal Analysis and Calorimetry. DOI: 10.1007/s10973-023-12439-z 2002. Enhanced enantioselectivity of lipase from Pseudomonas sp. at high temperatures and fixed water activity in the ionic liquid, 1-butyl-3-methylimidazolium bis[(trifluoromethyl)sulfonyl]amide. Biotechnology Letters. DOI: 10.1023/a:1015563801977 |
| Market Analysis Reports |