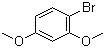
1-Bromo-2,4-dimethoxybenzene is an aromatic organic compound consisting of a benzene ring with bromine and two methoxy groups (–OCH3) attached to the 1 and 4 positions of the ring. This compound belongs to the family of halogenated methoxybenzenes and is a valuable intermediate in organic synthesis. It is of particular interest in the development of complex molecules for use in pharmaceuticals, agrochemicals, and materials science. The discovery of 1-bromo-2,4-dimethoxybenzene is closely tied to the study of halogenated aromatic compounds, which have long been explored for their reactivity and utility in various chemical reactions.
The synthesis of 1-bromo-2,4-dimethoxybenzene typically involves a selective bromination reaction using a suitable brominating agent, such as bromine or NBS (N-Bromosuccinimide), on 2,4-dimethoxyphenol or a similar precursor. The process introduces the bromine atom at the 1-position of the aromatic ring, ensuring that the methoxy groups remain intact at the 2 and 4 positions. This selective functionalization of the benzene ring is important for creating compounds with specific reactivity that can be further utilized in various synthetic transformations.
One of the primary applications of 1-bromo-2,4-dimethoxybenzene is in organic synthesis, where it acts as a key intermediate for constructing more complex chemical structures. The presence of both halogen and methoxy groups on the benzene ring allows for a variety of nucleophilic aromatic substitution reactions, enabling the creation of derivatives with diverse functional groups. These derivatives are useful for the development of bioactive compounds, especially in medicinal chemistry.
In the field of medicinal chemistry, 1-bromo-2,4-dimethoxybenzene has been employed in the synthesis of compounds with potential therapeutic applications. Its functionalized structure makes it an attractive precursor for the creation of molecules targeting specific biological pathways. For example, it has been used in the design of pharmaceuticals that possess anti-inflammatory, antimicrobial, or anticancer properties. The methoxy groups provide electron-donating effects that can enhance the solubility and bioavailability of the resulting drug candidates, improving their pharmacokinetic profiles.
1-Bromo-2,4-dimethoxybenzene also has applications in the agrochemical industry, where it is used to synthesize various pesticides and herbicides. The ability to introduce functional groups onto the aromatic ring allows for the development of compounds that target specific pests or plant diseases. The stability and selectivity of halogenated aromatics are key factors in enhancing the effectiveness of agrochemical products, and 1-bromo-2,4-dimethoxybenzene’s structure makes it a useful building block for this purpose.
In materials science, 1-bromo-2,4-dimethoxybenzene has been explored for its role in the synthesis of organic semiconductors and conductive polymers. The methoxy groups on the benzene ring can modify the electronic properties of the compound, making it suitable for use in organic electronics. This includes applications in the production of organic light-emitting diodes (OLEDs) and organic photovoltaic cells. The versatility of 1-bromo-2,4-dimethoxybenzene in forming derivatives with tailored properties contributes to its potential in these advanced technological applications.
Despite its promising applications, the use of 1-bromo-2,4-dimethoxybenzene must be approached with caution due to the environmental and safety concerns associated with halogenated compounds. Brominated aromatic compounds can pose environmental risks, and their disposal requires careful management to avoid contamination. As with all chemical synthesis, proper handling, storage, and waste disposal practices are necessary to mitigate the potential hazards of using such substances.
In conclusion, 1-bromo-2,4-dimethoxybenzene is a significant compound in organic synthesis with diverse applications in medicinal chemistry, agrochemicals, and materials science. Its unique combination of bromine and methoxy groups makes it a versatile intermediate for the development of bioactive compounds, agricultural chemicals, and organic materials. Ongoing research into its properties and potential applications continues to expand the scope of its use in various industrial and technological fields.
References
2007. A convenient and selective method for the para-bromination of aromatic compounds with potassium bromide in the presence of poly(4-vinylpyridine)-supported peroxodisulfate in nonaqueous solution. Russian Journal of Organic Chemistry, 43.
DOI: 10.1134/s1070428007090047
2021. Recent Advances in the Synthesis of Diverse Libraries of Small-Molecule Building Blocks in Ionic Liquids (ILs). Synlett, 32.
DOI: 10.1055/s-0040-1719852
2014. Application of bisphosphomide-palladium(II) pincer complex in Suzuki-Miyaura cross-coupling reaction under microwave irradiation. Journal of Chemical Sciences, 126.
DOI: 10.1007/s12039-014-0594-z
|































































 GHS07 Warning Details
GHS07 Warning Details