Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Suzhou Chireach Biomedical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chireach.com | |||
 | +86 (512) 6585-6968 +86 13862507957 | |||
 | +86 (512) 6564-7218 | |||
 | cathydai@chireach.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou TJM Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjmchem.com | |||
 | +86 13388471767 | |||
 | +86 (571) 8822-3279 | |||
 | sales@tjmchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shenzhen FCD Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fcdtech.com | |||
 | +86 18668616292 | |||
 | +86 18668616292 | |||
 | fcd-sd@fcdtech.com | |||
 | QQ Chat | |||
 | WeChat: +86 18668616292 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Xinyi Chemical Research Institute | China | |||
|---|---|---|---|---|
 | www.xinyichemical.com | |||
 | +86 (519) 8646-2478 8647-1022 | |||
 | +86 (519) 8646-8478 | |||
 | lxy.mq@163.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | Parylene F Dimer |
| Synonyms | 5,6,11,12,13,14,15,16-Octafluorotricyclo[8.2.2.24,7]hexadeca-4,6,10,12,13,15-hexaene |
| Molecular Structure | 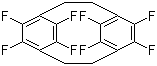 |
| Molecular Formula | C16H8F8 |
| Molecular Weight | 352.22 |
| CAS Registry Number | 1785-64-4 |
| SMILES | C1CC2=C(C(=C(CCC3=C(C(=C1C(=C3F)F)F)F)C(=C2F)F)F)F |
| Density | 1.497 |
|---|---|
| Boiling point | 321 °C |
| Flash point | 117 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Parylene F dimer, a key precursor for the formation of Parylene F polymer, is a member of the Parylene family, which consists of several types of chemical vapor deposition (CVD) polymers known for their exceptional chemical resistance, thermal stability, and electrical insulation properties. The chemical structure of Parylene F dimer features two fused benzene rings with fluorine atoms attached at specific positions, contributing to the polymer's high-performance characteristics. This substance has played a vital role in advancing materials science, particularly in the areas of protective coatings for sensitive electronics and biomedical devices. The discovery of Parylene F dimer builds on the original development of Parylene polymers by William F. Gorham in the late 1950s and early 1960s, during his time at Union Carbide Corporation. The first member of the Parylene family, Parylene N, was synthesized through a two-step pyrolysis process that involved the thermal cleavage of a dimer into reactive monomers, which then polymerized on surfaces via vapor deposition. The success of Parylene N led to the creation of other variants, such as Parylene C and Parylene F, each with modified chemical structures to enhance specific properties. Parylene F dimer, specifically, was developed to meet the growing demand for materials that could withstand extreme conditions, such as high temperatures and chemically aggressive environments. By introducing fluorine atoms into the structure of the dimer, the resulting polymer exhibited improved thermal and chemical stability compared to its predecessors. Parylene F can operate at temperatures exceeding 250°C and demonstrates resistance to many corrosive chemicals, making it ideal for applications that require durable protective coatings. The primary application of Parylene F dimer lies in its use as a precursor for the Parylene F polymer, which is deposited as an ultra-thin, conformal coating. This polymer is widely used in the electronics industry to protect delicate components from moisture, dust, chemicals, and other environmental factors that could compromise performance. Parylene F coatings are applied to circuit boards, sensors, and other microelectronic devices to enhance their longevity and reliability, particularly in harsh operating environments, such as aerospace, automotive, and oil exploration sectors. In addition to its role in electronics, Parylene F has found significant use in the medical field. Its biocompatibility, combined with its protective properties, makes it suitable for coating medical devices, such as catheters, stents, and pacemakers, where durability and non-reactivity with biological tissues are essential. The polymer's ability to form a pinhole-free barrier ensures that medical devices maintain their functionality while reducing the risk of contamination or failure in biological systems. Another important application of Parylene F is in the realm of photonics and optoelectronics, where its transparent nature in the visible and near-infrared spectra, along with its excellent dielectric properties, make it an ideal candidate for optical coatings. It is used in devices such as light-emitting diodes (LEDs), lasers, and sensors, where high-performance coatings are necessary to ensure optimal light transmission and electrical insulation. Parylene F dimer also serves as a critical material for research into advanced nanotechnology and surface science. The vapor deposition process used to create Parylene F films allows for the precise control of film thickness, making it useful in the fabrication of nanoscale devices and the protection of delicate structures at the molecular level. Researchers are exploring its potential applications in areas such as flexible electronics, wearable devices, and even biomedical implants that interface directly with the human body. The combination of thermal stability, chemical resistance, and biocompatibility provided by Parylene F dimer-derived coatings has made it an indispensable material in various industries. As technologies continue to advance, particularly in fields such as electronics, biotechnology, and nanotechnology, the applications of Parylene F are likely to expand further, driven by its unique properties and versatility as a high-performance protective coating. References 2009. Mild and efficient conversion of trifluoromethylarenes into tribromomethylarenes using boron tribromide. Chemical Papers. DOI: 10.2478/s11696-008-0082-3 2002. Vapor Deposition Polymerization as a Route to Fluorinated Polymers. Fluoropolymers 1: Synthesis. DOI: 10.1007/0-306-46918-9_18 1995. Vapor Deposition Of Very Low K Polymer Films, Poly(Naphthalene), Poly(Fluorinated Naphthalene). MRS Online Proceedings Library. DOI: 10.1557/proc-381-45 |
| Market Analysis Reports |