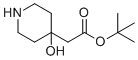
Tert-butyl 2-(4-hydroxypiperidin-4-yl)acetate is an organic compound composed of a substituted piperidine ring bearing a hydroxyl group at the 4-position, and a tert-butyl ester of acetic acid attached at the same carbon. This structure combines a six-membered saturated heterocycle with a sterically protected carboxylic acid derivative, offering functional flexibility for synthetic applications.
The piperidine ring is a common motif in medicinal chemistry, often valued for its conformational rigidity and ability to modulate bioavailability and binding affinity in pharmaceutical candidates. In this compound, the 4-position is doubly substituted—bearing both a hydroxyl group and a side chain leading to the ester. The presence of the tert-butyl ester protects the carboxylic acid from premature hydrolysis during synthetic transformations, allowing for downstream reactions without loss of the acid functionality.
Synthesis of tert-butyl 2-(4-hydroxypiperidin-4-yl)acetate generally begins with the functionalization of 4-hydroxypiperidine or protected piperidinone derivatives. Alkylation of the 4-position with tert-butyl bromoacetate or similar esters under basic conditions leads to the formation of the ester-substituted product. The reaction typically proceeds under mild conditions and is scalable for both research and preparative purposes.
This compound serves as a key intermediate in the synthesis of drug-like molecules, particularly those targeting central nervous system receptors or enzymes. The hydroxyl group at the 4-position provides a handle for further functionalization, including etherification, acylation, or conversion to leaving groups for substitution reactions. The tert-butyl ester can be removed under acidic conditions (e.g., trifluoroacetic acid) to reveal the corresponding carboxylic acid for subsequent amide coupling or salt formation.
Compounds bearing similar piperidine-based side chains have appeared in small-molecule inhibitors of proteases, GPCR ligands, and enzyme cofactors. The balance of hydrophilicity from the hydroxyl and carboxylic acid functionalities and the lipophilicity imparted by the tert-butyl group makes this compound suitable for optimizing pharmacokinetic properties in lead development programs.
In conclusion, tert-butyl 2-(4-hydroxypiperidin-4-yl)acetate is a synthetically useful intermediate that merges a functionalized piperidine core with a protected carboxylic acid moiety. Its modular design allows for diverse chemical modifications, making it a valuable building block in medicinal chemistry and drug discovery research.
|




 GHS07 Warning Details
GHS07 Warning Details