Online Database of Chemicals from Around the World
| TH Chemica Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thchemica.com | |||
 | +86 (573) 8283-5115 +86 13998887555 | |||
 | +86 (573) 8396-5652 | |||
 | thchemica@126.com info@thchemica.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Zhejiang Kaili Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjklsy.com | |||
 | +86 (571) 8524-1926 +86 18758150316 | |||
 | +86 (571) 8524-1907 | |||
 | sales@zjklsy.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| Endeavour Speciality Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.endeavourchem.co.uk | |||
 | +44 (1327) 310-079 | |||
 | +44 (1327) 310-701 | |||
 | enquiries@endeavourchem.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aryl compounds >> Anilines |
|---|---|
| Name | 2-(4-Methylpiperazin-1-yl)aniline |
| Molecular Structure | 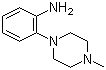 |
| Molecular Formula | C11H17N3 |
| Molecular Weight | 191.27 |
| CAS Registry Number | 180605-36-1 |
| EC Number | 641-312-2 |
| SMILES | CN1CCN(CC1)C2=CC=CC=C2N |
| Melting point | 101-102 °C |
|---|---|
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302+H312+H332-H302-H311-H312-H314-H318-H331-H332 Details | ||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P361+P364-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-(4-Methylpiperazin-1-yl)aniline is a chemical compound that has found significant applications in medicinal chemistry, particularly in the development of pharmaceutical agents. The compound is characterized by the presence of a piperazine ring, substituted with a methyl group, attached to an aniline moiety. This structural configuration imparts the molecule with both basicity and the potential for hydrogen bonding, making it a versatile intermediate in organic synthesis. The discovery of 2-(4-methylpiperazin-1-yl)aniline can be traced back to efforts in synthesizing heterocyclic compounds with enhanced pharmacological properties. The piperazine ring is a common motif in many drugs due to its ability to interact with biological targets, including receptors and enzymes. The introduction of the methyl group at the nitrogen atom of the piperazine ring and the aniline group enhances the compound's pharmacokinetic properties, such as solubility and membrane permeability, making it a valuable building block in drug discovery. One of the primary applications of 2-(4-methylpiperazin-1-yl)aniline is in the synthesis of antihistamines, antipsychotics, and antidepressants. The compound serves as a key intermediate in the production of these drugs, contributing to the pharmacological activity of the final products. For example, it is used in the synthesis of certain antipsychotic drugs, where it acts as a precursor to more complex molecules that target dopamine receptors in the brain. The ability of the piperazine ring to mimic natural neurotransmitters and bind to their respective receptors makes 2-(4-methylpiperazin-1-yl)aniline a critical component in the development of central nervous system (CNS) active drugs. In addition to its role in drug synthesis, 2-(4-methylpiperazin-1-yl)aniline is also used in the development of anticancer agents. The compound's structure allows it to be easily modified to create molecules that can inhibit the growth of cancer cells by targeting specific enzymes or signaling pathways involved in cell proliferation. This application has made 2-(4-methylpiperazin-1-yl)aniline a valuable tool in the ongoing search for new and effective cancer treatments. Researchers have explored its derivatives for their potential to improve the selectivity and potency of anticancer drugs, leading to the development of novel therapeutic agents. The versatility of 2-(4-methylpiperazin-1-yl)aniline extends beyond pharmaceuticals. The compound is also employed in the synthesis of agrochemicals, where it contributes to the development of pesticides and herbicides. Its ability to interact with biological systems allows it to be used in the formulation of compounds that target specific pests or weeds, reducing the environmental impact and improving the efficiency of agricultural practices. The compound's stability and reactivity make it an ideal candidate for creating durable and effective agrochemical agents. Research into 2-(4-methylpiperazin-1-yl)aniline continues to explore its potential in new areas of application. The compound's basicity and ability to form hydrogen bonds make it an attractive starting material for the synthesis of polymers and other materials with specialized properties. These materials could be used in various industries, including electronics, where they may serve as components in the manufacture of conductive polymers or as additives in the production of high-performance materials. The ongoing interest in 2-(4-methylpiperazin-1-yl)aniline is driven by its versatility and the ease with which it can be modified to suit different purposes. As new therapeutic targets and industrial applications are discovered, this compound is likely to play an increasingly important role in both medicinal chemistry and material science. The combination of its unique structural features and its broad applicability ensures that 2-(4-methylpiperazin-1-yl)aniline will remain a valuable chemical substance in a variety of scientific and industrial fields. References 2023. Open science discovery of potent noncovalent SARS-CoV-2 main protease inhibitors. Science (New York, N.Y.). DOI: 10.1126/science.abo7201 2023. Multimodal action of KRP203 on phosphoinositide kinases in vitro and in cells. Biochemical and Biophysical Research Communications. DOI: 10.1016/j.bbrc.2023.08.050 2015. Cell-based and virtual fragment screening for adrenergic α2C receptor agonists. Bioorganic & Medicinal Chemistry. DOI: 10.1016/j.bmc.2015.01.013 |
| Market Analysis Reports |