Online Database of Chemicals from Around the World
| Fujian Wolfa Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wolfabio.com | |||
 | +86 18050950397 | |||
 | jomin@wolfabio.com | |||
 | WhatsApp:+86 18359103607 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic scandium, tantalum, thallium, tungsten, antimony, lanthanum, lead, vanadium, molybdenum, chromium, ytterbium, etc. |
|---|---|
| Name | Triamminemolybdenum(0) tricarbonyl |
| Synonyms | azane carbon monoxide molybdenum |
| Molecular Structure | 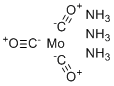 |
| Molecular Formula | C3H9MoN3O3 |
| Molecular Weight | 231.08 |
| CAS Registry Number | 18177-91-8 |
| EC Number | 622-401-5 |
| SMILES | [C-]#[O+].[C-]#[O+].[C-]#[O+].N.N.N.[Mo] |
| Melting point | 220-225 °C |
|---|---|
| Hazard Symbols |  GHS02 Danger Details GHS02 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H261 Details | ||||||||
| Safety Statements | P231+P232-P280-P370+P378-P402+P404-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Triamminemolybdenum(0) tricarbonyl is a notable organometallic compound with significant implications in chemical synthesis and catalysis. This compound is formed from the coordination of three carbonyl groups and an ammine ligand to a molybdenum center in its zero oxidation state. It exemplifies the diverse reactivity and utility of transition metal carbonyl complexes in various chemical processes. The discovery of triamminemolybdenum(0) tricarbonyl dates back to research aimed at understanding the behavior of molybdenum in its lower oxidation states. Transition metal carbonyl complexes, particularly those involving molybdenum, have been subjects of intense study due to their unique electronic properties and catalytic activities. Triamminemolybdenum(0) tricarbonyl was synthesized as part of this broader investigation into the reactivity of molybdenum carbonyls, providing insights into the metal's coordination chemistry and reactivity under different conditions. The synthesis of triamminemolybdenum(0) tricarbonyl typically involves the reaction of molybdenum hexacarbonyl with an ammine ligand. The process is carried out under controlled conditions to ensure the formation of the desired complex. The resulting compound features a molybdenum center coordinated to three carbonyl groups and an ammine ligand, providing a stable and well-defined structure for further study. In terms of applications, triamminemolybdenum(0) tricarbonyl is primarily used as a catalyst in various chemical reactions. Transition metal carbonyl complexes are renowned for their ability to facilitate a range of transformations, including hydrogenation, carbonylation, and olefin metathesis. Triamminemolybdenum(0) tricarbonyl is no exception and has been employed in reactions where its unique electronic configuration and coordination environment offer catalytic advantages. One prominent application of triamminemolybdenum(0) tricarbonyl is in the field of organic synthesis, where it serves as a catalyst for carbonylation reactions. These reactions involve the introduction of carbonyl groups into organic substrates, a process that is essential for the synthesis of various chemical products, including pharmaceuticals and agrochemicals. The presence of the carbonyl groups in the triamminemolybdenum(0) tricarbonyl complex facilitates the activation of carbon monoxide, thereby enhancing the efficiency of the carbonylation process. In addition to carbonylation, triamminemolybdenum(0) tricarbonyl is also utilized in the study of reaction mechanisms and the development of new catalytic processes. Its well-characterized structure and reactivity provide valuable information for understanding how transition metal carbonyl complexes interact with substrates and participate in chemical transformations. This knowledge is crucial for the design of more efficient catalysts and the optimization of reaction conditions in industrial processes. The compound's stability and solubility in various solvents make it a versatile reagent in laboratory research. Its use extends to academic studies aimed at exploring new catalytic strategies and understanding the fundamental principles of transition metal chemistry. In summary, triamminemolybdenum(0) tricarbonyl is an important organometallic compound with significant implications for catalysis and organic synthesis. Its discovery and subsequent applications highlight the role of transition metal carbonyl complexes in advancing chemical research and industrial processes. References none |
| Market Analysis Reports |