Chloroplatinic acid hexahydrate, commonly known as hexachloroplatinic acid hexahydrate and chemically represented as H2PtCl6·6H2O, is an important inorganic platinum compound. It consists of the hexachloroplatinate(IV) anion [PtCl6]2− paired with two protons (H+) and six molecules of water of crystallization. The compound typically forms as red-orange crystalline solids and is highly soluble in water, forming strongly acidic solutions.
Chloroplatinic acid was first prepared in the early 19th century following the isolation of platinum metal and its compounds. The compound is typically obtained by dissolving platinum metal or platinum alloys in aqua regia, a mixture of nitric and hydrochloric acids, which oxidizes the platinum and complexes it with chloride ions. Subsequent evaporation and crystallization from the aqueous solution yields the hexahydrate form.
Structurally, the hexachloroplatinate(IV) anion features a central platinum ion in the +4 oxidation state coordinated octahedrally by six chloride ligands. The strong Pt–Cl bonds and the octahedral symmetry confer significant stability to the complex. The two protons associate with water molecules and contribute to the acidic nature of the compound in solution.
Chloroplatinic acid hexahydrate serves as a crucial precursor in platinum chemistry and catalysis. It is widely used for the preparation of platinum-based catalysts through reduction or impregnation methods. These catalysts find extensive applications in hydrogenation, oxidation, and reforming reactions in the chemical and petrochemical industries. The compound’s ability to release platinum ions in a controlled manner facilitates the synthesis of supported platinum catalysts with high dispersion and activity.
In addition to catalysis, chloroplatinic acid is employed in the production of platinum metal by reduction processes. It is also used in electroplating to deposit thin, uniform platinum coatings on various substrates, providing corrosion resistance and catalytic properties.
The compound is a valuable reagent in analytical chemistry for platinum quantification and in materials science for fabricating platinum-containing films and nanoparticles. Its high solubility and well-defined chemistry make it suitable for these precise applications.
Handling chloroplatinic acid hexahydrate requires care due to its strong acidity and potential toxicity. It is corrosive and can cause severe burns upon contact with skin or eyes. Inhalation or ingestion poses health hazards. Appropriate safety precautions include using personal protective equipment, working in well-ventilated environments or fume hoods, and proper storage in corrosion-resistant containers.
Thermally, chloroplatinic acid hexahydrate decomposes upon heating to release hydrogen chloride and chlorine gases and leaves behind platinum oxides or metallic platinum depending on the conditions. Controlled decomposition is utilized in catalyst preparation and platinum recovery.
In summary, chloroplatinic acid hexahydrate is a water-soluble, strongly acidic platinum(IV) chloride complex widely used as a precursor for platinum catalysts, metal production, and electroplating. Its stable octahedral hexachloroplatinate(IV) anion, combined with high solubility and reactivity, underpins its extensive applications in industrial and laboratory platinum chemistry.
References
none
|














































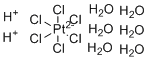


 GHS06;GHS08;GHS05 Danger Details
GHS06;GHS08;GHS05 Danger Details