Online Database of Chemicals from Around the World
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic base |
|---|---|
| Name | Cerium trichloride heptahydrate |
| Synonyms | Cerous chloride heptahydrate |
| Molecular Structure | 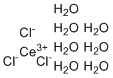 |
| Molecular Formula | CeCl3.7(H2O) |
| Molecular Weight | 372.59 |
| CAS Registry Number | 18618-55-8 |
| EC Number | 606-073-0 |
| SMILES | O.O.O.O.O.O.O.[Cl-].[Cl-].[Cl-].[Ce+3] |
| Density | ~3.94 g/mL (25 °C) |
|---|---|
| Melting point | 103 °C |
| Hazard Symbols |   GHS05;GHS09 Danger Details GHS05;GHS09 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H410 Details | ||||||||||||||||
| Safety Statements | P260-P273-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Cerium trichloride itself has been known since the early 19th century, when Martin Heinrich Klaproth and Jöns Jacob Berzelius discovered cerium in 1803. The hydrate, cerium trichloride heptahydrate, has been extensively studied as researchers explored the chemical properties of rare earth elements and their salts. The main feature of the chemical structure is that the central cerium ion (Ce³⁺) is coordinated by three chloride ions. Each cerium trichloride unit is associated with seven water molecules, which affects its solubility and reactivity. It is a colorless to pale yellow crystal that decomposes at 100-110°C, losing its hydration water. It is highly soluble in water, forming a clear solution; it is also soluble in ethanol and methanol. Cerium trichloride heptahydrate is widely used as a Lewis acid in organic synthesis. Its ability to accept electron pairs makes it an effective catalyst for a variety of reactions, including Diels-Alder reactions, Friedel-Crafts acylations, and Michael addition reactions. It facilitates the addition of Grignard reagents to carbonyl compounds, thereby increasing the yield and selectivity of the desired product. This is particularly useful in forming carbon-carbon bonds in complex organic molecules. CeO₂ heptahydrate is a precursor for the preparation of cerium dioxide (CeO₂), a material widely used in catalysis, polishing agents, and solid oxide fuel cells. The hydration state affects the morphology and properties of the resulting cerium dioxide. It is used in the synthesis of cerium-containing nanomaterials for catalysis, luminescence, and as an antioxidant in biomedical research. In environmental chemistry, cerium chloride heptahydrate is used in catalytic processes to degrade pollutants, such as the catalytic reduction of nitrogen oxides (NOx) and the oxidation of organic pollutants. It is used as a catalyst in fuel additives to improve combustion efficiency and reduce emissions, especially in diesel engines. The compound helps to reduce the activation energy required for the oxidation of fuel components. CeO₂ heptahydrate is used to form coordination complexes with various ligands. Studies of the structural and electronic properties of these complexes have helped to understand lanthanide chemistry. Its complexes are also valuable in spectroscopic studies, which provide insights into the behavior of cerium ions in different chemical environments. The compound is used as a reagent in analytical chemistry to detect and quantify a variety of substances. Its reactivity with both organic and inorganic analytes makes it a useful tool for laboratory analysis. References 2024. Synergistic effect of cerium chloride and calcium chloride alters calcium signaling in keratinocytes to promote epidermal differentiation. Bioscience, Biotechnology, and Biochemistry. DOI: 10.1093/bbb/zbae131 2017. Cerium chloride stimulated controlled conversion of B-to-Z DNA in self-assembled nanostructures. Biochemical and Biophysical Research Communications. DOI: 10.1016/j.bbrc.2016.11.133 2012. CeCl3·7H2O Catalyzed C-C and C-N Bond-Forming Cascade Cyclization with Subsequent Side-Chain Functionalization and Rearrangement: A Domino Approach to Pentasubstituted Pyrrole Analogues. The Journal of Organic Chemistry. DOI: 10.1021/jo301796r |
| Market Analysis Reports |