Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Utopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.utopharm.com | |||
 | +86 (21) 5409-1930 +86 13816330796 | |||
 | +86 (21) 5409-1296 | |||
 | uto2010008@utopharm.com utopharm8@yahoo.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shijiazhuang Lida Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lid-ff.cn | |||
 | +86 (311) 8573-7996 | |||
 | +86 (311) 8573-9175 | |||
 | service@lid-ff.cn | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jingjiang Connect Chemical Manufacturing Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.katchemical.com | |||
 | +86 (523) 8050-1690 +86 15252623102 | |||
 | +86 (523) 8450-6952 | |||
 | andy@katchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2012 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Zibo Kunran Enterprises Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kunranchem.com | |||
 | +86 (533) 520-0667 | |||
 | +86 (533) 520-7669 | |||
 | monazhangchem@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Missyou Chenmical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shmychem.com | |||
 | +86 (21) 5858-3907 | |||
 | +86 (21) 5858-3907 | |||
 | doreen@shmychem.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Huai'an Huaitai Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huataichem.com | |||
 | +86 (517) 8599-9999 | |||
 | +86 (517) 8532-2222 | |||
 | sales@huataichem.com | |||
| Chemical manufacturer | ||||
| DeLong Chemicals America | USA | |||
|---|---|---|---|---|
 | www.delongchemicals.com | |||
 | +1 (203) 271-9017 | |||
 | sales@delongchemicals.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 2-(2-Bromoethyl)-1,3-dioxolane |
| Synonyms | 3-Bromopropionaldehyde ethylene acetal |
| Molecular Structure | 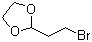 |
| Molecular Formula | C5H9BrO2 |
| Molecular Weight | 181.03 |
| CAS Registry Number | 18742-02-4 |
| EC Number | 242-551-1 |
| SMILES | C1COC(O1)CCBr |
| Density | 1.5±0.1 g/cm3 Calc.*, 1.512 g/mL (Expl.) |
|---|---|
| Boiling point | 200.9±15.0 °C 760 mmHg (Calc.)*, 214.7 - 217.5 °C (Expl.) |
| Flash point | 85.0 °C (Calc.)*, 65 °C (Expl.) |
| Solubility | water: insoluble (Expl.) |
| Index of refraction | 1.473 (Calc.)*, 1.479 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
2‑(2‑Bromoethyl)‑1,3‑dioxolane is a halogenated acetal featuring a five‑membered 1,3‑dioxolane ring substituted at the 2‑position by a bromoethyl side chain Its molecular structure combines an electrophilic bromide and a carbonyl‑protecting acetal ring making it a multifunctional reagent in organic synthesis The bromide moiety undergoes clean nucleophilic substitution reactions with amines thiols azides or organometallic reagents allowing introduction of protected hydroxyethyl or other substituents The acetal ring offers orthogonal protection of aldehyde or ketone functionalities and can be cleaved under mild acidic conditions regenerating reactive carbonyl groups for downstream transformations This compound is typically prepared by bromination of acetals such as acrolein‑ethylene glycol derivatives under controlled conditions using hydrogen bromide in a solvent such as ethyl acetate or 1,4‑dioxane Recent synthetic developments have improved yield and purity and simplified reagent handling making it more accessible for research and development applications The dual reactivity of the compound enables tandem sequences such as acetal deprotection and immediate functionalization of the released carbonyl or alkylation followed by cleavage in multistep protocols In medicinal chemistry the compound has been explored as an intermediate to build substituted heterocycles and natural product analogues polymer precursors and as a linker in click chemistry applications In materials science it can serve to introduce protected functional groups onto polymer backbones through halide‑mediated cross‑coupling or click reactions The stability profile under ambient conditions paired with selective reactivity makes it valuable in both solution phase and automated synthesis workflows Due to its brominated alkyl group and acetal protection standard safety precautions are required to prevent alkylation or acid hydrolysis Its environmental and toxicological properties align with other bromoalkyl acetals which are typically irritants and require care during handling References Christensen J B (1994) An economical preparation of 2‑(2‑bromoethyl)‑1,3‑dioxolane Org Prep Proced Int 26 3 355–357 DOI: 10.1080/00304949409458436 Nishio Y Mifune R Sato T Ishikawa S Matsubara H (2017) Hydrogen bromide in 1,4‑dioxane a controlled source for bromination of acrolein acetals Tetrahedron Lett 58 12 1190–1193 DOI: 10.1016/j.tetlet.2017.02.020 Ponaras A A (2001) 2‑(2‑Bromoethyl)‑1,3‑dioxolane Encyclopedia of Reagents for Organic Synthesis John Wiley & Sons Ltd rb291 DOI:10.1002/047084289X.rb291 |
| Market Analysis Reports |