Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Wuxi Orient Detergent Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinobromide.com | |||
 | +86 (510) 8758-1290 | |||
 | +86 (510) 8758-3694 | |||
 | suguangming@126.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Yancheng Longshen Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longshenchem.com | |||
 | +86 (515) 8773-1896 +86 18261286666 | |||
 | +86 (515) 8865-2999 | |||
 | 2355756927@qq.com gaojunwyt@vip.163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Anhui Weixiang New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ahwxchem.com | |||
 | +86 13195623283 | |||
 | export@ahwxchem.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| FAR Chemical, Inc. | USA | |||
|---|---|---|---|---|
 | www.far-chemical.com | |||
 | +1 (321) 723-6160 | |||
 | +1 (321) 723-8753 | |||
 | info@far-chemical.com | |||
| Chemical manufacturer since 1983 | ||||
| Yancheng Creator Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.creatorchem.com | |||
 | +86 (515) 8871-0008 8871-0868 8307-2966 | |||
 | +86 (515) 8871-0858 / 8307-0966 | |||
 | yc@jssdchem.com | |||
| Chemical manufacturer | ||||
| Sanmenxia Aoke Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aokeby.com | |||
 | +86 (398) 383-4379 | |||
 | +86 (398) 383-0625 | |||
 | h99139@hotmail.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| ChemSampCo, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemsampco.com | |||
 | +1 (609) 656-2440 | |||
 | +1 (609) 656-2446 | |||
 | sales@chemsampco.com | |||
| Chemical manufacturer since 1960 | ||||
| Classification | Chemical reagent >> Organic reagent >> Halogenated aliphatic hydrocarbon |
|---|---|
| Name | 2-Ethylhexyl bromide |
| Synonyms | 1-Bromo-2-ethylhexane; 3-(Bromomethyl)heptane |
| Molecular Structure | 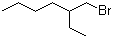 |
| Molecular Formula | C8H17Br |
| Molecular Weight | 193.13 |
| CAS Registry Number | 18908-66-2 |
| EC Number | 242-659-9 |
| SMILES | CCCCC(CC)CBr |
| Density | 1.1±0.1 g/cm3 Calc.*, 1.086 g/mL (Expl.) |
|---|---|
| Boiling point | 190.8±8.0 °C 760 mmHg (Calc.)*, 75-77 °°C (16 mmHg) (Expl.) |
| Flash point | 69.4 °C (Calc.)*, 69 °C (Expl.) |
| Solubility | water insoluble (Expl.) |
| Index of refraction | 1.45 (Calc.)*, 1.453-1.455 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-Ethylhexyl bromide is a branched primary alkyl halide with the molecular formula C8H17Br. It appears as a colorless to pale yellow liquid and is widely recognized for its utility as an intermediate in organic synthesis. The compound features a bromine atom attached to the terminal carbon of a 2-ethylhexyl chain, which confers strong electrophilic properties, making it highly reactive toward nucleophiles and suitable for substitution reactions. The synthesis of 2-ethylhexyl bromide commonly involves the reaction of 2-ethylhexanol with hydrobromic acid, phosphorus tribromide, or other brominating agents. This transformation typically proceeds via a substitution mechanism, converting the hydroxyl group into a bromide with retention of the carbon skeleton. Industrial production emphasizes high yield and selectivity to minimize side reactions such as elimination, which can produce alkenes. Chemically, 2-ethylhexyl bromide serves as a versatile intermediate due to the bromide leaving group. It readily participates in SN2 reactions, allowing substitution with nucleophiles including alcohols, amines, thiols, and carboxylates to form ethers, esters, amines, and other functionalized derivatives. Its branched structure reduces the likelihood of elimination compared with linear analogs, which makes it particularly valuable in selective synthetic applications. One of the most common applications of 2-ethylhexyl bromide is in the preparation of plasticizers. It is a key precursor for 2-ethylhexyl esters, such as 2-ethylhexyl phthalate and other similar compounds used to impart flexibility and durability to plastics. The compound also finds use in the synthesis of surfactants, lubricants, and specialty chemicals where the introduction of a branched alkyl chain can modify solubility, hydrophobicity, or other physicochemical properties. In the pharmaceutical and agrochemical industries, 2-ethylhexyl bromide acts as a building block for more complex molecules. Its bromide functionality allows for the introduction of a variety of substituents at the terminal carbon, enabling chemists to design molecules with specific biological or chemical properties. This reactivity under controlled conditions allows for the production of compounds used in drug formulation, crop protection, and other specialty applications. From a mechanistic standpoint, 2-ethylhexyl bromide is an important model for studying nucleophilic substitution reactions in branched systems. Its reactivity profile provides insight into steric effects and electronic influences on reaction kinetics, which helps chemists optimize conditions for efficient synthesis. The branched 2-ethylhexyl group also contributes to altered physical properties of resulting derivatives, influencing solubility, volatility, and compatibility in various formulations. Physically, 2-ethylhexyl bromide is insoluble in water but miscible with common organic solvents such as alcohols, ethers, and hydrocarbons. It has a moderate boiling point and exhibits low vapor pressure relative to smaller haloalkanes, which facilitates handling in industrial and laboratory environments. Safety measures are essential due to its irritant properties and potential toxicity. Protective gloves, eye protection, and proper ventilation are required to minimize exposure, and environmental precautions are important to prevent release into water systems, as it can be harmful to aquatic life. The discovery and widespread use of 2-ethylhexyl bromide underscore its importance as a flexible and reactive intermediate in organic chemistry. Its ability to undergo selective nucleophilic substitution reactions, combined with the properties conferred by its branched chain, ensures its continued relevance in industrial, pharmaceutical, and research applications. References 2016. Quantitative analysis of amphiphilic N-alkyloxypyridinecarboximidamide by liquid chromatography-tandem mass spectrometry. Chemical Papers, 71(6). DOI: 10.1007/s11696-016-0019-1 2021. Studies on Amino Acid Type Protic Ionic Liquid Comprising N-2-Ethylhexylethylenediaminium Cation Coupled with the dl-Hexanoylalaninate Anion. Journal of Solution Chemistry, 50(6). DOI: 10.1007/s10953-021-01097-0 2017. Reaction of Arene- or Methanesulfonates with Lithium Bromide in Tetrahydrofuran. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-135-00221 |
| Market Analysis Reports |