Online Database of Chemicals from Around the World
| Wuhan Silworld Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.silworldchemical.com | |||
 | +86 (27) 8561-3400 +86 15827173649 | |||
 | +86 (27) 8561-3419 | |||
 | info@silworldchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hubei Lansun Biochemistry Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lansunpharma.com | |||
 | +86 (714) 639-5977 | |||
 | +86 (714) 639-5877 | |||
 | info@lansunpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hubei Sinem Flavor Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinem.cn | |||
 | +86 (27) 8818-9660 | |||
 | sales@sinem.cn | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty ketone (including enol) |
|---|---|
| Name | (E)-4-Phenyl-3-buten-2-one |
| Synonyms | trans-4-Phenyl-3-buten-2-one; trans-Benzalacetone; trans-Benzylideneacetone; BAR |
| Molecular Structure | 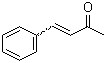 |
| Molecular Formula | C10H10O |
| Molecular Weight | 146.19 |
| CAS Registry Number | 1896-62-4 |
| EC Number | 217-587-6 |
| FEMA | 2881 |
| SMILES | CC(=O)/C=C/C1=CC=CC=C1 |
| Water solubility | practically insoluble |
|---|---|
| Density | 1.0±0.1 g/mL, Calc.*, 1.038 g/mL (Expl.) |
| Melting point | 39-42 °C (Expl.) |
| Index of Refraction | 1.563, Calc.* |
| Boiling Point | 260.8±9.0 °C (760 mmHg), Calc.*, 260-262 °C (Expl.) |
| Flash Point | 65.6 °C, Calc.*, 123 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H317-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P272-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(E)-4-Phenyl-3-buten-2-one, also known as (E)-cinnamyl ketone, is an organic compound that belongs to the family of chalcones. It is characterized by the presence of a phenyl group attached to a conjugated alkene-ketone structure. This compound is typically produced through the aldol condensation reaction, where a phenylacetaldehyde reacts with acetone in the presence of a suitable catalyst. The discovery of (E)-4-phenyl-3-buten-2-one dates back to early synthetic organic chemistry studies aimed at understanding conjugated systems and their reactivity. It has since been identified as a key intermediate in the synthesis of various natural and synthetic organic compounds. The main applications of (E)-4-phenyl-3-buten-2-one are found in the fragrance and flavor industries. It has a sweet, floral, and somewhat spicy aroma, making it a valuable component in the formulation of perfumes, soaps, and cosmetic products. In the food industry, it is used as a flavoring agent to impart cinnamon-like or spicy notes to various products, including confectionery and baked goods. Additionally, (E)-4-phenyl-3-buten-2-one has been studied for its potential use as an intermediate in the synthesis of other organic compounds, including pharmaceuticals and agrochemicals. It is also of interest in the field of materials science for its incorporation into polymers and other functional materials. While the compound has not been extensively studied for its medicinal properties, its aromatic profile and chemical reactivity suggest potential uses in diverse chemical synthesis processes. It is generally considered to have low toxicity, though care should be taken when handling the compound, especially in concentrated forms. In conclusion, (E)-4-phenyl-3-buten-2-one is a valuable compound in the fragrance, flavor, and chemical industries, with its sweet-spicy aroma contributing to its widespread use. Its role as a synthetic intermediate in organic chemistry further underscores its importance in chemical synthesis. References 2023. Multimodal action of KRP203 on phosphoinositide kinases in vitro and in cells. Biochemical and Biophysical Research Communications, . DOI: 10.1016/j.bbrc.2023.08.050 2024. Progress in the Secondary Metabolite Production in Medicinal Plants: An Overview. Biosynthesis of Natural Products in Plants, . DOI: 10.1007/978-981-97-2166-5_17 |
| Market Analysis Reports |