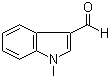
1-Methylindole-3-carboxaldehyde is an organic compound that consists of an indole core structure, with a methyl group attached to the nitrogen atom at position 1 of the indole ring, and an aldehyde functional group at the 3-position of the ring. This compound is a derivative of indole, a heterocyclic aromatic organic compound known for its presence in various natural products, including alkaloids and plant metabolites.
The discovery of 1-methylindole-3-carboxaldehyde is linked to the broader field of indole chemistry, which has been studied extensively due to the structural versatility and bioactivity of indole derivatives. The indole nucleus serves as the foundation for a variety of bioactive compounds, many of which have pharmaceutical or agricultural applications. The methylation at position 1 of the indole ring introduces a substitution that can influence the compound's chemical and physical properties, as well as its reactivity in synthetic processes.
One of the main applications of 1-methylindole-3-carboxaldehyde lies in its role as a synthetic intermediate. The presence of the aldehyde group at the 3-position makes this compound highly reactive, allowing it to undergo nucleophilic addition reactions, condensation reactions, or other transformations to form more complex structures. This reactivity is valuable for creating a wide range of indole-based derivatives, which can be used as building blocks in the synthesis of pharmaceuticals, agrochemicals, and other organic compounds.
In particular, 1-methylindole-3-carboxaldehyde and its derivatives have been explored in the development of compounds with antimicrobial, anti-inflammatory, and anticancer activities. The indole scaffold is well-known for its presence in biologically active molecules, and the introduction of functional groups such as aldehydes can enhance or modify the biological properties of these compounds. For example, compounds derived from 1-methylindole-3-carboxaldehyde have been studied for their potential to inhibit enzymes or interact with cellular targets involved in various disease processes.
Additionally, 1-methylindole-3-carboxaldehyde can be utilized in the synthesis of natural product derivatives. For instance, the indole ring system is found in several naturally occurring compounds, including tryptophan, serotonin, and various alkaloids, many of which exhibit significant biological activity. By modifying the indole ring with different substituents like the aldehyde group, chemists can design new molecules with potentially enhanced or novel activities for use in therapeutic applications.
In materials science, 1-methylindole-3-carboxaldehyde and its derivatives have also been considered for their potential use in the development of organic semiconductors. The indole ring has conjugated π-electron systems, which can contribute to the electronic properties of materials. As a result, this compound and its derivatives are of interest in the field of organic electronics, particularly in the development of organic light-emitting diodes (OLEDs) and organic photovoltaic cells (OPVs), where the molecular design of materials plays a critical role in their performance.
The compound's reactivity also makes it valuable for use in cross-coupling reactions, such as the Suzuki, Heck, or Sonogashira coupling, to form new carbon-carbon bonds. These reactions are essential in synthetic chemistry for the preparation of complex organic molecules. In particular, the ability to functionalize the indole ring through these reactions opens up new avenues for the synthesis of diverse chemical structures with applications in drug discovery, materials science, and other fields.
In conclusion, 1-methylindole-3-carboxaldehyde is a significant compound in organic synthesis due to its reactive aldehyde group and the indole backbone. Its applications span medicinal chemistry, materials science, and synthetic organic chemistry. As a versatile intermediate, it plays an important role in the development of new biologically active compounds and novel materials with potential applications in various industries.
References
2001. 1-Methylindole-3-carboxaldehyde oxime derivatives. Acta Crystallographica Section C: Crystal Structure Communications, 57(1).
DOI: 10.1107/s0108270100005795
2019. Symmetrical and unsymmetrical azomethines with thiophene core: structure-properties investigations. Journal of Materials Science, 54(24).
DOI: 10.1007/s10853-019-03853-6
2017. Formation of C-Carbon (Alkenylation). Science of Synthesis, 1.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-110-01021
|







































 GHS07 Warning Details
GHS07 Warning Details