Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Benzyl 2,2-dimethylbut-3-ynoate |
| Molecular Structure | 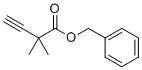 |
| Molecular Formula | C13H14O2 |
| Molecular Weight | 202.25 |
| CAS Registry Number | 204588-77-2 |
| SMILES | CC(C)(C#C)C(=O)OCC1=CC=CC=C1 |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.521, Calc.* |
| Boiling Point | 280.4±23.0 °C (760 mmHg), Calc.* |
| Flash Point | 112.6±20.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P264-P270-P301+P312-P330 Details |
| SDS | Available |
|
Benzyl 2,2-dimethylbut-3-ynoate is an organic compound with a distinctive ester structure, containing a benzyl group attached to the ester functionality along with a dimethyl-substituted alkyne chain. The molecular formula of this compound is C12H14O2. It is characterized by a benzyl ester attached to a 2,2-dimethylbut-3-yne moiety, making it a valuable intermediate in organic synthesis. The initial discovery and synthesis of benzyl 2,2-dimethylbut-3-ynoate occurred in research focused on the development of functionalized alkynes, a class of compounds prized for their versatile reactivity and use as building blocks in chemical synthesis. One of the main applications of benzyl 2,2-dimethylbut-3-ynoate is in synthetic organic chemistry, where it is used as a building block for the synthesis of complex molecules. The presence of the terminal alkyne and benzyl ester group enables this compound to participate in a wide range of chemical reactions, including nucleophilic additions, coupling reactions, and ester hydrolysis. These properties make it useful in the design of advanced organic frameworks, especially in the pharmaceutical industry, where such frameworks are integral to drug discovery and development. In addition to its use as a synthetic intermediate, benzyl 2,2-dimethylbut-3-ynoate has shown potential in the synthesis of bioactive molecules and materials with unique properties. Its reactive alkyne group allows it to participate in reactions such as the Sonogashira and Suzuki coupling reactions, which are commonly employed to introduce aromatic or heterocyclic moieties into complex molecular structures. This makes the compound particularly valuable in the creation of pharmacologically active compounds, including inhibitors, enzyme substrates, and molecular probes. Recent research has also focused on benzyl 2,2-dimethylbut-3-ynoate’s role in material science. The alkyne functionality allows for the creation of polymers and dendrimers, where the ester linkage provides controlled degradability. This dual functionality is beneficial for developing responsive materials and biodegradable polymers, leading to applications in fields such as biomedicine and environmental science. Overall, benzyl 2,2-dimethylbut-3-ynoate is an important compound with diverse applications in organic synthesis, pharmaceuticals, and material science. Its chemical versatility and functional groups enable its widespread use as a building block in creating complex molecules, high-value materials, and innovative pharmaceutical agents, supporting further advancements across multiple disciplines. References 2018. Tezacaftor. Pharmaceutical Substances. https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-20-0240 |
| Market Analysis Reports |