Online Database of Chemicals from Around the World
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal chlorides and salts |
|---|---|
| Name | Manganese(II) chloride dihydrate |
| Synonyms | Manganese dichloride dihydrate; Manganous chloride dihydrate |
| Molecular Structure | 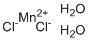 |
| Molecular Formula | MnCl2.2(H2O) |
| Molecular Weight | 161.87 |
| CAS Registry Number | 20603-88-7 |
| EC Number | 682-147-6 |
| SMILES | O.O.[Cl-].[Cl-].[Mn+2] |
| Hazard Symbols |    GHS06;GHS07;GHS09 Danger Details GHS06;GHS07;GHS09 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H411 Details | ||||||||||||||||
| Safety Statements | P264-P270-P273-P301+P316-P301+P317-P321-P330-P391-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Manganese(II) chloride dihydrate, with the chemical formula MnCl2·2H2O, is an inorganic compound that plays a significant role in various chemical, industrial, and biological processes. The discovery of manganese(II) chloride can be traced back to the early studies of manganese compounds in the 18th century when chemists first identified manganese as a distinct element. However, the isolation of manganese(II) chloride specifically occurred later as researchers sought to understand the properties and applications of this metal salt. Manganese(II) chloride dihydrate is typically synthesized through the reaction of manganese(II) oxide or manganese(II) carbonate with hydrochloric acid. The resulting compound forms pale pink to light brown crystals that are soluble in water. Its dihydrate form indicates the presence of two water molecules per formula unit, which contributes to its stability and solubility in aqueous solutions. One of the primary applications of manganese(II) chloride dihydrate is in the field of fertilizers. Manganese is an essential micronutrient for plants, playing a crucial role in photosynthesis, nitrogen assimilation, and the synthesis of various enzymes. Manganese(II) chloride is often included in fertilizer formulations to ensure adequate manganese supply for crops, thus promoting healthy growth and enhancing yield. In addition to its agricultural uses, manganese(II) chloride dihydrate is widely employed in the chemical industry. It serves as a precursor for the synthesis of other manganese compounds, including manganese dioxide and manganese sulfate. These compounds find applications in the production of batteries, ceramics, and glass. Manganese(II) chloride is also utilized in the preparation of catalysts for organic reactions, particularly in oxidation processes. The compound is important in the field of analytical chemistry as well. It is commonly used as a reagent in various analytical techniques, such as titrations and colorimetric analyses. Its ability to form colored complexes with certain anions and ligands enables its use in determining the concentration of various substances in solution. Moreover, manganese(II) chloride dihydrate has applications in the field of biochemistry and medicine. It is involved in the study of manganese-dependent enzymes, which are crucial for various metabolic processes in living organisms. Additionally, research has indicated that manganese compounds, including manganese(II) chloride, may have therapeutic potential in certain medical conditions, particularly in the context of neurological health. Despite its benefits, the handling of manganese(II) chloride dihydrate requires caution due to potential toxicity associated with manganese exposure. While manganese is an essential trace element, excessive intake can lead to health issues, including neurological disorders. Therefore, safety measures must be adhered to in both industrial and laboratory settings to minimize risks. In summary, manganese(II) chloride dihydrate is a vital chemical compound with diverse applications ranging from agriculture to chemical synthesis and biochemistry. Its discovery and subsequent use underscore the importance of manganese in various fields, emphasizing the need for continued research and responsible handling of this important compound. References 2024. Manganese Overexposure Alters Neurogranin Expression and Causes Behavioral Deficits in Larval Zebrafish. International Journal of Molecular Sciences, 25(9). DOI: 10.3390/ijms25094933 2021. Manganese (II) chloride leads to dopaminergic neurotoxicity by promoting mitophagy through BNIP3-mediated oxidative stress in SH-SY5Y cells. Cellular & Molecular Biology Letters, 26(1). DOI: 10.1186/s11658-021-00267-8 1951. MUTAGENIC ACTION OF MANGANOUS CHLORIDE. Cold Spring Harbor Symposia on Quantitative Biology, 16. DOI: 10.1101/sqb.1951.016.01.017 |
| Market Analysis Reports |