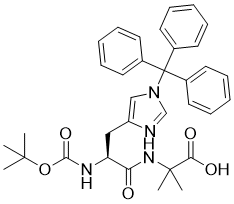
Boc-His(Trt)-Aib-OH is a synthetic peptide building block used in solid-phase peptide synthesis (SPPS). This compound plays a significant role in the design and development of peptides with therapeutic potential, as well as in studying protein structure and function. The chemical structure of Boc-His(Trt)-Aib-OH includes three main components: Boc (tert-butyloxycarbonyl) as a protecting group, Trt (trityl) for the selective protection of the imidazole side chain of histidine, and Aib (α-aminoisobutyric acid), a non-natural amino acid with unique conformational properties.
The discovery of Boc-His(Trt)-Aib-OH is rooted in the advancement of peptide synthesis techniques, specifically SPPS, pioneered by Robert Bruce Merrifield in the 1960s. This method revolutionized peptide chemistry by enabling the assembly of peptides on a solid resin, allowing for the stepwise addition of amino acids. Protecting groups like Boc and Trt were developed to prevent unwanted side reactions during synthesis. Boc protects the amino group of histidine, while Trt protects the imidazole side chain, ensuring that selective reactions can take place at specific functional sites.
Boc-His(Trt)-Aib-OH is primarily used in the synthesis of peptides that contain the histidine residue and Aib. Histidine, an essential amino acid, is involved in many biological processes, including enzyme catalysis and metal ion coordination. Aib, on the other hand, imparts conformational rigidity due to its steric hindrance, which influences peptide structure by promoting helical conformations. This property makes Aib-containing peptides highly valuable in the study of protein folding and stability.
The selective use of Boc-His(Trt)-Aib-OH in peptide synthesis has applications in various fields, including drug discovery and material science. Therapeutic peptides, often used to target specific molecular pathways in diseases such as cancer and metabolic disorders, rely on precise peptide sequences. The inclusion of Aib in these sequences helps to enhance the stability and bioavailability of the peptides, making them more effective as drugs. Additionally, Boc-His(Trt)-Aib-OH is used in the design of peptide-based materials for biomedical applications, such as hydrogels and nanostructures, where the structural properties of Aib-containing peptides contribute to the mechanical and functional attributes of the materials.
Research on Boc-His(Trt)-Aib-OH continues to explore its utility in producing peptides with improved pharmacokinetics and stability. Its role in SPPS makes it an essential tool for the development of peptides with diverse biological activities, from enzyme inhibitors to antimicrobial agents. The compound’s combination of selective protecting groups and the unique properties of Aib make it indispensable in modern peptide chemistry.
References
none
|











 GHS07 Warning Details
GHS07 Warning Details