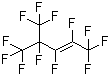
Perfluoro(4-methylpent-2-ene) is a fluorinated organic compound belonging to the class of perfluoroalkenes, characterized by the substitution of all hydrogen atoms in a hydrocarbon chain with fluorine atoms. Its molecular structure consists of a six-carbon backbone with a double bond at the second position and a methyl group at the fourth carbon, all fully fluorinated. The resulting chemical formula is C6F12, and the compound is known for its thermal and chemical stability, as well as its high density and low surface energy—properties commonly associated with perfluorinated substances.
The discovery and development of perfluorinated compounds such as perfluoro(4-methylpent-2-ene) began in the mid-20th century, primarily through research conducted by chemical companies exploring the use of fluorine in organic chemistry. Perfluorinated olefins attracted interest due to their potential utility as intermediates in the synthesis of fluoropolymers, surfactants, and other specialized materials. Companies like 3M and DuPont were instrumental in pioneering fluorine chemistry, leading to the discovery and commercial exploitation of numerous perfluorinated chemicals.
Perfluoro(4-methylpent-2-ene) has been investigated as a reactive intermediate in the synthesis of fluorinated polymers and surface treatment agents. The double bond in its molecular structure allows it to undergo addition reactions, making it useful for chemical modifications, especially in controlled polymerization processes. Such reactions can lead to the formation of highly fluorinated side-chains or crosslinked networks with applications in coatings, sealants, and membranes.
One of the notable applications of this compound is in the production of fluorosurfactants and surface-active agents. Due to its strong carbon–fluorine bonds and the resulting low polarizability and surface energy, derivatives of perfluoro(4-methylpent-2-ene) can impart water- and oil-repellency to treated surfaces. These properties are highly desirable in the manufacture of stain-resistant textiles, non-stick cookware coatings, and protective films.
In addition, perfluoro(4-methylpent-2-ene) and related compounds have been studied as monomers in the preparation of fluoroelastomers and fluorinated polymers. These materials are prized for their resistance to aggressive chemicals, solvents, and high temperatures. In industrial settings, they are used in applications such as gaskets, seals, and linings in chemical processing equipment.
Another significant area of application involves the use of perfluorinated compounds in electronic and aerospace industries, where materials are required to withstand extreme environmental conditions while maintaining electrical insulation or structural integrity. Polymers and surface coatings derived from compounds like perfluoro(4-methylpent-2-ene) can provide durable, low-friction, and non-reactive barriers for sensitive components.
While the compound itself is not commonly used in consumer products, its chemical characteristics make it valuable in niche industrial processes where performance under harsh conditions is essential. However, like many perfluorinated substances, it may present environmental and toxicological concerns. Perfluorinated compounds are known for their persistence in the environment, resistance to degradation, and potential for bioaccumulation. As such, their use and disposal are subject to increasing regulatory scrutiny.
Modern research into perfluoroalkenes continues to explore their reactivity and potential for novel materials, particularly in the design of fluorinated block copolymers and other advanced macromolecular architectures. These efforts aim to balance the desirable physicochemical properties of perfluorinated compounds with considerations for environmental safety and regulatory compliance.
In conclusion, perfluoro(4-methylpent-2-ene) is a fluorinated olefinic compound valued for its stability and reactivity in the synthesis of high-performance materials. It exemplifies the broader class of perfluorinated compounds, which have contributed significantly to advances in materials science, especially in contexts requiring extreme resistance to chemical, thermal, or environmental degradation.
References
1989 Formation of perfluoro-1,1,3-trimethylindane in the reaction of trimethylsilylpentafluorobenzene with perfluoro-4-methyl-2-pentene. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 38(2).
DOI: 10.1007/bf00953645
1991 Unexpected formation of an α-hydrazone in the reaction of perfluoro-4-methyl-2-pentene with hydrazine hydrate. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 40(6).
DOI: 10.1007/bf00961321
2012 From Fluoroalkenes. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-127-00266
|










































 GHS07 Warning Details
GHS07 Warning Details