Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Daji Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.djpharm.com | |||
 | +86 13816245196 | |||
 | sales1@djpharm.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Rintech, Inc. | USA | |||
|---|---|---|---|---|
 | www.rintechinc.com | |||
 | +1 (301) 987-1980 | |||
 | +1 (301) 987-1981 | |||
 | info@rintechinc.com | |||
| Chemical manufacturer since 1997 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Ketone compound |
|---|---|
| Name | Isochroman-4-one |
| Synonyms | 1H-isochromen-4-one |
| Molecular Structure | 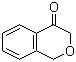 |
| Molecular Formula | C9H8O2 |
| Molecular Weight | 148.16 |
| CAS Registry Number | 20924-56-5 |
| EC Number | 819-563-6 |
| SMILES | C1C2=CC=CC=C2C(=O)CO1 |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 279.5±19.0 °C 760 mmHg (Calc.)* |
| Flash point | 134.7±15.1 °C (Calc.)* |
| Index of refraction | 1.562 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Isochroman-4-one is a bicyclic heterocyclic compound consisting of a benzene ring fused to a saturated oxygen-containing six-membered ring, forming the isochroman scaffold, with a ketone functional group at position 4. Its molecular formula is C9H8O2, and it has a molecular weight of approximately 148.16 g/mol. The fused bicyclic system imparts rigidity and planarity to the molecule, while the ketone at position 4 introduces electrophilic character and reactivity toward nucleophiles. The compound is generally synthesized through cyclization reactions that form the oxygen-containing ring fused to the aromatic core. One common method involves intramolecular acylation of o-alkylated benzoic acid derivatives, where the side-chain carbonyl reacts under acidic or basic catalysis to form the lactone structure. Alternative approaches include oxidation of isochroman derivatives or selective cyclization of hydroxybenzyl precursors, providing flexibility in the introduction of substituents or isotopic labels. Reaction conditions are typically chosen to favor formation of the six-membered oxygen-containing ring and to avoid over-oxidation or polymerization of the aromatic system. Chemically, isochroman-4-one is moderately polar due to the carbonyl group and the ether oxygen within the ring. The ketone at position 4 is susceptible to nucleophilic attack, enabling reactions such as reduction to the corresponding alcohol, formation of imines or hydrazones, and enolate chemistry. The oxygen atom in the heterocyclic ring also affects the electronic distribution in the molecule, influencing both aromatic electrophilic substitution and the acidity of the hydrogen atoms adjacent to the carbonyl group. In organic synthesis, isochroman-4-one is used as a versatile intermediate for the preparation of more complex heterocycles and substituted derivatives. The ketone allows selective functionalization at position 4, while the aromatic core can undergo electrophilic substitution reactions. The fused ring system provides a rigid framework that is valuable in the design of bioactive molecules and natural product analogs, enabling spatially defined substitution patterns that can influence molecular recognition or binding properties. The compound is generally a solid at room temperature and exhibits moderate solubility in polar organic solvents such as ethanol, acetone, or dichloromethane. Its stability under ambient conditions is adequate for routine laboratory handling, though it should be protected from strong oxidizing agents and nucleophiles that could react with the ketone or ether functionalities. The planar aromatic system contributes to crystallinity, which can be useful for purification by recrystallization or for structural characterization. In practical applications, isochroman-4-one serves as a building block in medicinal chemistry and materials science. Its structural features—combining a fused aromatic-oxygen heterocycle with a reactive ketone—enable the preparation of biologically relevant analogs, chromophores, and intermediates for polymeric materials. Its ability to undergo diverse chemical transformations while maintaining the integrity of the fused ring makes it a flexible intermediate for synthetic strategies aimed at generating heterocyclic libraries or functionalized derivatives. References 2023. Role of Computational Modeling in Drug Discovery for Alzheimer’s Disease. Challenges and Advances in Computational Chemistry and Physics. DOI: 10.1007/978-3-031-33871-7_3 2017. Cytotoxic tropolones from the fungus Nemania sp. BCC 30850. Tetrahedron. DOI: 10.1016/j.tet.2017.05.030 2015. Facile Approach for C(sp3)–H Bond Thioetherification of Isochroman. Synlett. DOI: 10.1055/s-0034-1380125 |
| Market Analysis Reports |