Online Database of Chemicals from Around the World
| Targetmol Chemicals Inc. | USA | |||
|---|---|---|---|---|
 | www.targetmol.com | |||
 | +1 (781) 999-5354 | |||
 | +1 (781) 281-9145 | |||
 | sales@targetmol.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Organic fluorine compound |
|---|---|
| Name | PFB-2G-Fluorescein |
| Synonyms | 2,3,4,5,6-pentafluoro-N-[3-oxo-3',6'-bis[[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy]spiro[2-benzofuran-1,9'-xanthene]-5-yl]benzamide |
| Molecular Structure | 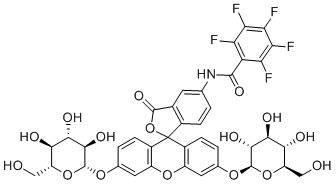 |
| Molecular Formula | C39H32F5NO16 |
| Molecular Weight | 865.66 |
| CAS Registry Number | 209540-62-5 |
| SMILES | C1=CC2=C(C=C1NC(=O)C3=C(C(=C(C(=C3F)F)F)F)F)C(=O)OC24C5=C(C=C(C=C5)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O)OC7=C4C=CC(=C7)O[C@H]8[C@@H]([C@H]([C@@H]([C@H](O8)CO)O)O)O |
| Density | 1.8±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 1012.2±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 565.9±34.3 °C (Calc.)* |
| Index of refraction | 1.727 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details |
| SDS | Available |
|
PFB-2G-Fluorescein is a fluorescein-based compound widely used in the field of biochemical and cellular analysis, particularly in applications involving fluorescence labeling and detection. It is a derivative of fluorescein, a well-known xanthene dye characterized by its strong fluorescence and utility in various labeling and tracing techniques. The structure of PFB-2G-Fluorescein incorporates two glucose (2G) moieties and a pentafluorobenzyl (PFB) group, which modify its chemical properties to enhance solubility, targeting, or chemical reactivity depending on the application. Fluorescein itself was first synthesized in the 19th century and quickly became a cornerstone in the development of fluorescent dyes due to its high quantum yield and excitation/emission properties that are compatible with standard fluorescence detection systems. The core structure of fluorescein allows for chemical modification at several positions, enabling the design of derivatives with tailored characteristics for specific analytical tasks. PFB-2G-Fluorescein was developed as one such derivative to meet specialized analytical demands, particularly in the realm of mass spectrometry and bioanalytical chemistry. The incorporation of a pentafluorobenzyl group is especially relevant in derivatization chemistry for gas chromatography-mass spectrometry (GC-MS). The PFB moiety enhances electron-capturing capability, making PFB-derivatives highly suitable for negative ion chemical ionization (NICI) detection, which improves sensitivity and selectivity. In the case of PFB-2G-Fluorescein, the electron-capturing nature of the pentafluorobenzyl group enables its use in analytical workflows that require high sensitivity in detecting labeled compounds. This makes it suitable for the analysis of amino acids, peptides, carbohydrates, and other biomolecules that may be derivatized for detection. The two glucose residues (2G) contribute to increased hydrophilicity and may also play a role in targeting or solubility in biological systems. Glycosylated fluorescein derivatives can enhance compatibility with aqueous biological media and can be used in labeling protocols that involve living cells or tissues. In some cases, glycosylation may also affect cellular uptake or localization, although the specific targeting characteristics of PFB-2G-Fluorescein are determined by its complete molecular context and application. PFB-2G-Fluorescein has found use in experimental workflows involving fluorescence-based detection and quantification, as well as mass spectrometric assays requiring derivatization of analytes. It can be applied to label molecules through suitable reactive functional groups introduced into the compound, allowing it to bind covalently to amines, carboxyls, thiols, or hydroxyl groups, depending on the derivatization chemistry employed. This versatility makes it a useful reagent for proteomics, glycomics, and other molecular profiling techniques. While the compound is primarily used in research settings, its utility reflects broader trends in the development of multifunctional fluorescent reagents that combine chemical reactivity, analytical sensitivity, and biological compatibility. The design of such compounds supports the advancement of detection technologies in modern biochemical and clinical laboratories. PFB-2G-Fluorescein continues to be a part of the growing suite of fluorescent tags used in experimental science to trace, quantify, and visualize molecular species with precision. References none |
| Market Analysis Reports |