Online Database of Chemicals from Around the World
| Changzhou Fluoride Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoridechem.com | |||
 | +86 (519) 8989-2883 +86 15995021858 | |||
 | +86 (519) 8522-7531 | |||
 | ycyvip@gmail.com ycyvip@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Novasyn Organics Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.novasynorganics.com | |||
 | +91 (406) 620-2233 633-9111 | |||
 | +91 (402) 781-1997 | |||
 | info@novasynorganics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| HetCat - Heterocycles & Catalysts | Switzerland | |||
|---|---|---|---|---|
 | www.hetcat.com | |||
 | +41 (61) 363-9868 | |||
 | +41 (61) 363-9869 | |||
 | fallahpour@hetcat.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| AB Chem, Inc. | Canada | |||
|---|---|---|---|---|
 | www.abcheminc.com | |||
 | +1 (514) 685-8688 | |||
 | +1 (514) 685-8488 | |||
 | sales@abcheminc.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Nitropyridine |
|---|---|
| Name | 2-Methyl-5-nitropyridine |
| Synonyms | 5-Nitro-2-picoline |
| Molecular Structure | 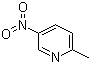 |
| Molecular Formula | C6H6N2O2 |
| Molecular Weight | 138.12 |
| CAS Registry Number | 21203-68-9 |
| EC Number | 677-568-7 |
| SMILES | CC1=NC=C(C=C1)[N+](=O)[O-] |
| Melting point | 107-109 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302+H312+H332-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2-Methyl-5-nitropyridine, a derivative of pyridine, was discovered in the mid-20th century during investigations into the reactivity and synthesis of pyridine compounds. The synthesis typically involves the nitration of 2-methylpyridine, resulting in the introduction of a nitro group at the 5-position of the pyridine ring. Its discovery marked a significant advancement in organic chemistry, providing chemists with a versatile building block. 2-Methyl-5-nitropyridine serves as a valuable intermediate in the synthesis of pharmaceutical compounds. Its unique structure and reactivity make it a versatile building block for the production of various drugs, including antihypertensives, antivirals, and anti-inflammatory agents. Chemical modifications of the nitro group or the pyridine ring enable the creation of drug candidates with improved efficacy, bioavailability, or selectivity. In the agricultural sector, 2-Methyl-5-nitropyridine finds application as a precursor for the synthesis of agrochemicals and pesticides. Pyridine derivatives possess pesticidal properties, and modifications of their structure can enhance their effectiveness against pests and weeds. By incorporating 2-Methyl-5-nitropyridine into pesticide formulations, researchers aim to develop products that can protect crops from damage caused by insects, fungi, and other pathogens. 2-Methyl-5-nitropyridine is valuable as an intermediate in organic synthesis, facilitating the production of various chemical compounds with diverse applications. Its incorporation into multistep synthesis pathways enables the creation of complex molecules used in pharmaceuticals, agrochemicals, and materials science. In laboratory settings, 2-Methyl-5-nitropyridine serves as a solvent or reagent in chemical reactions and synthesis processes. Its solubility properties and compatibility with a wide range of organic and inorganic compounds make it useful for dissolving or reacting with other substances to form desired products. Beyond its established applications, ongoing research explores new uses for 2-Methyl-5-nitropyridine in fields such as material science, catalysis, and drug discovery. Scientists investigate its reactivity, properties, and potential interactions with other molecules to develop innovative materials, catalysts, and chemical processes. References 2000. IR spectra of 2-alkylamino-and alkylnitramino-3-or 5-nitro-4-methylpyridine derivatives. Chemistry of Heterocyclic Compounds, 36(7). DOI: 10.1007/bf02256912 2016. Nitration of Pyridines. Science of Synthesis. URL: SD-115-00302 2012. Palladium-Catalyzed Asymmetric Addition of Alkylazaarenes to Imines and Nitroalkenes. Synfacts, 8(12). DOI: 10.1055/s-0032-1317750 |
| Market Analysis Reports |