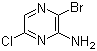
3-Bromo-6-chloro-2-pyrazinamine is a chemical compound belonging to the class of halogenated pyrazine derivatives. Pyrazine, a heterocyclic aromatic compound with two nitrogen atoms in the ring, serves as a core structure in this compound. The presence of the bromine and chlorine substituents at the 3- and 6-positions, respectively, on the pyrazine ring, significantly modifies its chemical properties, making it an interesting molecule for various applications in chemical synthesis and potentially in medicinal chemistry.
The specific positioning of the halogen atoms on the pyrazine ring suggests that the compound may exhibit unique electronic properties, as the electronegativity of chlorine and bromine can influence the reactivity of the compound. These halogen atoms can also provide sites for further functionalization, which is a valuable feature in synthetic chemistry, especially for the development of more complex molecules or for tuning the properties of the compound for specific applications.
The amine group at the 2-position on the pyrazine ring adds to the versatility of the compound, potentially allowing for interactions with other chemical species, including those in biological systems. The nitrogen atom in the amine group can act as a nucleophile, making the compound useful in various reactions, including those that form carbon-nitrogen bonds, a common strategy in organic synthesis.
This compound's halogenated structure, specifically the combination of bromine and chlorine, may contribute to its reactivity in organic reactions, making it a useful intermediate in the synthesis of other compounds. Additionally, the halogens may provide enhanced stability or reactivity under certain conditions, which could be beneficial for specific applications such as cross-coupling reactions or in the synthesis of more complex molecules.
In the context of medicinal chemistry, halogenated pyrazine derivatives are known for their bioactivity, including antimicrobial, anticancer, and anti-inflammatory properties. While direct data on the biological activities of 3-bromo-6-chloro-2-pyrazinamine is not readily available, compounds with similar structures have been explored for their potential to interact with various biological targets, such as enzymes or receptors, in the central nervous system or other organ systems. The presence of the amine group in particular could make it a potential candidate for drug development, as amines are often involved in interactions with biological macromolecules.
In summary, 3-bromo-6-chloro-2-pyrazinamine is a halogenated pyrazine derivative with potential applications in organic synthesis and medicinal chemistry. Its halogen-substituted structure may influence its reactivity and stability, making it a versatile intermediate in the preparation of more complex molecules. Additionally, its potential biological activity, driven by the unique combination of halogens and the amine group, could open up avenues for further exploration in drug discovery and development.
|




























 GHS07 Warning Details
GHS07 Warning Details