Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Alcohol |
|---|---|
| Name | trans-2,3-Dibromo-2-butene-1,4-diol |
| Synonyms | (E)-2,3-dibromobut-2-ene-1,4-diol |
| Molecular Structure | 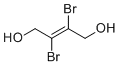 |
| Molecular Formula | C4H6Br2O2 |
| Molecular Weight | 245.90 |
| CAS Registry Number | 21285-46-1 |
| EC Number | 244-313-2 |
| SMILES | C(/C(=C(/CO)Br)/Br)O |
| Density | 2.3±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 114 °C (Expl.) |
| Index of Refraction | 1.630, Calc.* |
| Boiling Point | 318.1±42.0 °C (760 mmHg), Calc.* |
| Flash Point | 146.2±27.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317 Details | ||||||||
| Safety Statements | P261-P272-P280-P302+P352-P321-P333+P317-P362+P364-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
trans-2,3-Dibromo-2-butene-1,4-diol is an organic compound with a complex structure characterized by a butene backbone substituted with bromine atoms and hydroxyl groups. The compound was first synthesized in the mid-20th century as part of studies aimed at understanding the reactivity and properties of brominated organic compounds. Its unique structure positions it as an intriguing subject for both academic research and potential industrial applications. One of the primary applications of trans-2,3-dibromo-2-butene-1,4-diol is in the field of organic synthesis, where it serves as an important intermediate. The presence of bromine and hydroxyl functional groups allows for various chemical transformations, making it a versatile building block for the synthesis of more complex molecules. For example, the bromine atoms can be replaced through nucleophilic substitution reactions, leading to the formation of new carbon-carbon bonds and enabling the construction of diverse organic frameworks. This property is particularly useful in the development of pharmaceuticals, agrochemicals, and other fine chemicals. Additionally, trans-2,3-dibromo-2-butene-1,4-diol has been investigated for its potential biological activity. Preliminary studies have indicated that brominated compounds can exhibit antimicrobial properties, and research has explored the compound’s efficacy against certain bacteria and fungi. This aspect highlights the potential for developing new antimicrobial agents derived from trans-2,3-dibromo-2-butene-1,4-diol, particularly in the context of increasing antibiotic resistance. By modifying the structure of the compound, researchers aim to enhance its biological activity and specificity, potentially leading to the discovery of novel therapeutics. Moreover, the compound has been used in studies focusing on the environmental impact of brominated organic substances. Research has shown that certain brominated compounds can have significant effects on ecological systems, prompting investigations into their degradation pathways and toxicological effects. Understanding the environmental behavior of trans-2,3-dibromo-2-butene-1,4-diol can inform risk assessments and regulatory decisions concerning brominated compounds, particularly in the context of environmental pollution and sustainability. Overall, trans-2,3-dibromo-2-butene-1,4-diol represents a valuable compound in organic chemistry and medicinal research. Its applications in synthesis, potential biological activity, and environmental studies underscore its importance in advancing both scientific knowledge and practical applications in various fields. References 2012. Brominated Methanes as Photoresponsive Molecular Storage of Elemental Br2. Chemistry - An Asian Journal, 7(7). DOI: 10.1002/asia.201200322 2008. Addition of Bromine to Alkynes. Science of Synthesis. URL: SD-032-00100 |
| Market Analysis Reports |