Online Database of Chemicals from Around the World
| Chizhou Wanwei Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.www-chem.com | |||
 | +86 (566) 816-7432 +86 13696639666 | |||
 | +86 (566) 816-7550 | |||
 | zulwj@vip.163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai CK International Trade Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sh-chuo.com | |||
 | +86 (21) 6119-9292 6119-9293 +86 13311786807 | |||
 | +86 (21) 6119-9293 | |||
 | y-zhang@sh-chuo.com 13311786807@189.cn | |||
 | QQ Chat | |||
| Chemical distributor since 2002 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Deborn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shdeborn.com | |||
 | +86 (21) 6896-2123 +86 18017879299 | |||
 | +86 (21) 6896-3669 | |||
 | sales@shdeborn.com victorcheng@263.net | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Eustar Functional Material Technology, Inc. | China | |||
|---|---|---|---|---|
 | www.eustar.com.cn | |||
 | +86 (0512) 5510-5882 | |||
 | purchaser@eustar.com.cn | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Suzhou Ke Shengtong New Materials Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kstochina.com | |||
 | +86 (512) 6670-6833 | |||
 | +86 (512) 6670-6833 | |||
 | service@kstochina.com | |||
| Chemical manufacturer since 2016 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Rare Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.rarechem.de | |||
 | +49 (431) 5606-600 | |||
 | +49 (431) 5606-608 | |||
 | info@rarechem.de | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Chemical reagent >> Organic reagent >> Imide |
|---|---|
| Name | N,N'-Methanediylidenebis(2,6-diisopropylaniline) |
| Synonyms | Bis(2,6-diisopropylphenyl)carbodiimide |
| Molecular Structure | 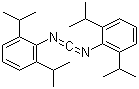 |
| Molecular Formula | C25H34N2 |
| Molecular Weight | 362.56 |
| CAS Registry Number | 2162-74-5 |
| EC Number | 218-487-5 |
| SMILES | CC(C)C1=C(C(=CC=C1)C(C)C)N=C=NC2=C(C=CC=C2C(C)C)C(C)C |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.529, Calc.* |
| Boiling Point | 477.7±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 235.6±29.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H360-H372 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P264-P270-P280-P301+P317-P318-P319-P330-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
N,N'-Methanediylidenebis(2,6-diisopropylaniline), commonly referred to as a Schiff base compound, is an organonitrogen compound notable for its versatile applications in coordination chemistry, polymer science, and industrial catalysis. This compound features two imine (-C=N-) linkages connecting a methylene group to two 2,6-diisopropylaniline units. Its molecular design combines steric hindrance and electron-donating properties, making it highly effective in stabilizing reactive intermediates and forming metal complexes. The discovery of Schiff bases dates back to the 19th century when Hugo Schiff identified the condensation reaction between primary amines and carbonyl compounds. N,N'-Methanediylidenebis(2,6-diisopropylaniline) emerged as part of efforts to create ligands with tailored electronic and steric properties. By incorporating 2,6-diisopropylaniline, researchers enhanced the compound's thermal stability, solubility, and selectivity for complexation with transition metals. This compound finds primary application as a ligand in the synthesis of metal complexes. Its bulky isopropyl groups help create spatially demanding environments around the metal center, enabling precise control over catalytic activity. For example, it is employed in olefin polymerization, where its complexes with titanium or zirconium act as highly efficient catalysts for producing polyethylene and polypropylene with tailored molecular weights and structures. In the realm of materials science, N,N'-Methanediylidenebis(2,6-diisopropylaniline) contributes to the development of advanced polymers. Its incorporation as a cross-linking agent or curing agent in epoxy resins enhances mechanical properties, thermal stability, and resistance to chemical degradation. These properties are crucial in applications ranging from aerospace components to high-performance adhesives and coatings. The compound also plays a role in dye and pigment synthesis, where its imine functionality participates in reactions to create chromophores with specific optical properties. Furthermore, its derivatives are studied for their potential in medical applications, such as drug delivery systems and as antimicrobial agents, leveraging its ability to bind selectively to biological targets. Environmental considerations are increasingly shaping its use, especially concerning sustainable catalysis and green chemistry. Research is directed toward designing recyclable catalysts based on its complexes and optimizing reaction conditions to minimize waste and energy consumption. N,N'-Methanediylidenebis(2,6-diisopropylaniline) illustrates the interplay between structural design and functionality, showcasing how tailored organic compounds can address diverse industrial and scientific challenges. Its applications span traditional and emerging fields, highlighting its adaptability and enduring relevance. References 2008. Synthesis and structure of two new (guanidinate)boron dichlorides and their attempted conversion to boron(i) derivatives. Dalton Transactions, (37). DOI: 10.1039/b800625c 2010. Steric control of the reduction of carbodiimides by samarium(II) and the synthesis of very crowded samarium(III) complexes. Dalton Transactions, 39(29). DOI: 10.1039/b922031c 2012. Studies on the reactivity of group 15 Zintl ions with carbodiimides: synthesis and characterization of a heptaphosphaguanidine dianion. Chemical Communications, 48(11). DOI: 10.1039/c1cc12089a |
| Market Analysis Reports |