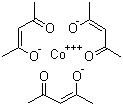
Cobaltic acetylacetonate is a coordination compound formed by the complexation of cobalt in its +3 oxidation state with acetylacetonate ligands. The acetylacetonate ligand, a bidentate chelating agent derived from acetylacetone, binds to the cobalt center through two oxygen atoms, forming stable five-membered chelate rings. This complex is often represented by the formula Co(acac)3, where three acetylacetonate ligands coordinate to a central cobalt ion.
The discovery of cobaltic acetylacetonate dates back to early coordination chemistry research focusing on transition metal complexes with β-diketone ligands. Acetylacetonate ligands gained prominence due to their ability to stabilize metal ions in various oxidation states, facilitating the isolation and study of metal complexes. Cobaltic acetylacetonate was characterized as a low-spin octahedral complex with cobalt in the +3 oxidation state, exhibiting distinctive electronic and magnetic properties. Its synthesis typically involves the oxidation of cobalt(II) precursors in the presence of acetylacetone under controlled conditions, leading to the formation of the cobalt(III) complex.
Cobaltic acetylacetonate is valued in coordination chemistry due to its stability, solubility in organic solvents, and well-defined structural features. It has been extensively studied using spectroscopic techniques such as UV-visible absorption, infrared spectroscopy, and nuclear magnetic resonance to elucidate its bonding and electronic configuration. The complex exhibits characteristic colors arising from d-d electronic transitions and ligand-to-metal charge transfer, which have been employed to study the electronic environments of cobalt centers.
Applications of cobaltic acetylacetonate primarily lie in the fields of catalysis, materials science, and organometallic chemistry. It serves as a precursor for the synthesis of cobalt-containing catalysts used in oxidation reactions and polymerization processes. The compound’s ability to undergo ligand substitution and redox reactions allows for the development of tailored catalytic systems. Additionally, cobaltic acetylacetonate is utilized in the preparation of thin films and coatings through techniques such as chemical vapor deposition, where it acts as a source of cobalt atoms.
In material science, cobaltic acetylacetonate has been investigated as a component in the fabrication of magnetic and electronic materials. Its paramagnetic properties and coordination environment influence the magnetic behavior of cobalt-based compounds derived from it. Furthermore, it finds use in fundamental studies of electron transfer processes and coordination dynamics in transition metal complexes.
Overall, cobaltic acetylacetonate represents a well-characterized cobalt(III) coordination complex with significant roles in coordination chemistry research and practical applications in catalysis and material fabrication. Its discovery contributed to the understanding of β-diketone ligand coordination and the chemistry of cobalt in higher oxidation states, providing a versatile platform for further chemical exploration.
References
none
|



























 GHS07;GHS08 Danger Details
GHS07;GHS08 Danger Details