Diallyldisulfide is an organosulfur compound that belongs to the class of disulfides and is naturally found in garlic (*Allium sativum*). Its molecular formula is C6H10S2, and it consists of two allyl groups (–CH2CH=CH2) linked via a disulfide bond (–S–S–). It is one of the major volatile compounds produced when garlic is crushed or processed and is largely responsible for garlic’s distinctive aroma and several of its biological activities.
Diallyldisulfide was first identified in the early 20th century during studies on garlic’s chemical composition. It is formed through the enzymatic breakdown of allicin, the primary bioactive compound in garlic, which itself is produced from alliin by the action of the enzyme alliinase when garlic tissues are damaged. Allicin is unstable and rapidly decomposes into various sulfur-containing compounds, including diallyldisulfide, diallyl trisulfide, and other polysulfides.
In terms of applications, diallyldisulfide has been studied extensively for its biological effects, particularly its antimicrobial, antifungal, antioxidant, and anticancer properties. In vitro and in vivo studies have demonstrated that it can inhibit the growth of various pathogenic microorganisms and may interfere with cellular mechanisms involved in cancer progression. Its mode of action includes the modulation of cellular signaling pathways, induction of apoptosis, and inhibition of angiogenesis. These activities are attributed to the electrophilic nature of the disulfide bond, which can interact with thiol-containing biomolecules such as glutathione or cysteine residues in proteins.
In agricultural and food science, diallyldisulfide has been evaluated as a natural pesticide and plant protectant due to its bioactivity against fungi, nematodes, and bacteria. Its volatile nature allows it to act as a fumigant in soil or storage environments. Additionally, it contributes to the flavor profile of garlic and garlic-derived products and is sometimes used as a food additive or flavoring agent in small concentrations.
Despite its promising biological properties, the practical use of diallyldisulfide in pharmaceuticals or commercial products has been limited by factors such as volatility, strong odor, and potential toxicity at high doses. Animal studies have indicated that high levels of exposure may cause irritation or toxicity in the liver and gastrointestinal tract. However, in dietary amounts from garlic consumption, it is generally considered safe and contributes to the health benefits associated with garlic intake.
In chemical synthesis, diallyldisulfide serves as a building block or intermediate in the preparation of sulfur-containing organic compounds. Its reactive disulfide bond can participate in various organic reactions, including radical or nucleophilic substitution, making it a useful reagent in synthetic organic chemistry.
In summary, diallyldisulfide is a naturally occurring disulfide compound derived from garlic, known for its pungent odor and broad spectrum of biological activity. It plays a role in the pharmacological effects of garlic and has been studied for its antimicrobial, antioxidant, and anticancer properties, while also finding uses in agriculture, flavoring, and chemical synthesis.
References
2016. DADS Analogues Ameliorated the Cognitive Impairments of Alzheimer-Like Rat Model Induced by Scopolamine. Neurotoxicity Research.
DOI: 10.1007/s12640-016-9625-5
2009. Diallyl disulfide induces apoptosis in human colon cancer cell line (COLO 205) through the induction of reactive oxygen species, endoplasmic reticulum stress, caspases casade and mitochondrial-dependent pathways. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 47(1).
DOI: 10.1016/j.fct.2008.10.032
2005. Diallyl disulfide inhibits N-acetyltransferase activity and gene expression in human esophagus epidermoid carcinoma CE 81T/VGH cells. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 43(7).
DOI: 10.1016/j.fct.2005.02.009
|

























































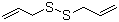


 GHS02;GHS06;GHS07 Danger Details
GHS02;GHS06;GHS07 Danger Details